Introduction: The intrinsic cardiac autonomic nervous system comprises interconnected epicardial ganglionated plexuses (GP) that are implicated in the initiation of atrial fibrillation (AF). GPs can be stimulated and functionally identified by delivering high frequency stimulation (HFS) within the local atrial refractory period, which can cause atrial ectopy and AF. These GPs are called ’ectopy-triggering GP‘ (ET-GP).
Objective: To identify the immunohistochemical characteristics underlying an ectopy-triggering response with GP stimulation.
Method: Four porcine hearts (4–5 months, 70–80 kg) were Langendorff-perfused. A 3.5 mm tip quadripolar ablation catheter was used for epicardial pacing at a fixed rate, and HFS was delivered within the local atrial refractory period (20 ms delay from pacing stimulus, 40 Hz, 10 V, 80 ms duration) using a custom-built stimulator. We tested globally around the left and right atria. Transmural dissection was performed at sites that induced ectopy or AF (ET-GP) and at sites with negative HFS response. We examined the tissues by brightfield microscopy, and by immunohistochemistry for identification of parasympathetic and sympathetic nerves, staining positive for choline acetyl transferase and tyrosine hydroxylase respectively. Immunohistochemistry on porcine sympathetic trunk and vagus nerve acted as positive controls.
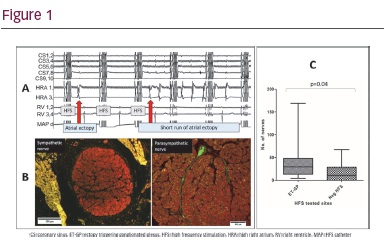
Results: Sixty-three HFS sites were tested, identifying 6 ET-GPs (10%) that evoked single ectopics and short runs of ectopy (Figure 1A). Six negative HFS sites were also analysed as controls. Sympathetic and parasympathetic nerves (Figure 1B) were identified in all ET-GP sites. One negative HFS site had no identifiable nerves. There were greater numbers of nerves in
ET-GPs compared with negative HFS sites (41 ± 44 versus 16 ± 20, p=0.04; Figure 1C). The sympathetic nerve cross-sectional area was smaller in
ET-GP sites compared with negative HFS (6,021µm2 ± 14,586 versus
22,786 µm2 ± 56,588, p<0.0001), though not significantly different for parasympathetic nerves (1,356 µm2 ± 2,127 versus 3,827 µm2 ± 10,156, p=0.06). We observed no differences in nerve distribution across the epicardium, myocardium or endocardium in both groups.
Conclusions: HFS can reproducibly trigger atrial ectopy in Langendorff-perfused porcine hearts. Ectopy-triggering sites have significantly larger numbers of sympathetic nerves that are smaller in size than in negative HFS sites. These local differences in sympathetic and parasympathetic nerve distribution and quantity may account for the differential functional response to HFS.