Patients with hypertrophic cardiomyopathy (HCM) are at risk of lethal ventricular arrhythmia. The electrical substrate for this has not been well elucidated. Furthermore, despite extensive knowledge of the structural differences in HCM vs the normal heart, the effects of electrophysiology are not known.
Methods: HCM patients surviving ventricular fibrillation or haemodynamically unstable ventricular tachycardia (HCM VF, n=17) were compared with HCM patients without a personal history of potentially lethal arrhythmia (HCM, n=20) and a pooled control group with structurally normal hearts but a range of conditions – 10 ischaemic cardiac arrest survivors with full revascularization and normalization of ventricular function, 11 patients undergoing ablation for benign VE and 11 patients proven to have normal hearts following family screening for Brugada syndrome. These 69 patients underwent exercise testing wearing a 252-electrode vest (CardioInsight ECGi, Medtronic) and a low-resolution CT. Activation time (AT) and activation recovery intervals (ARI) were compared between subjects to determine the electrical substrate in HCM.
Results: Most surface ECG markers were similar between the patient groups. QRS duration at both peak exercise and recovery, as well as QTc at peak exercise were not significantly different. The HCM VF group had a longer QTc in recovery than any group aside from the VE patients (p<0.01).
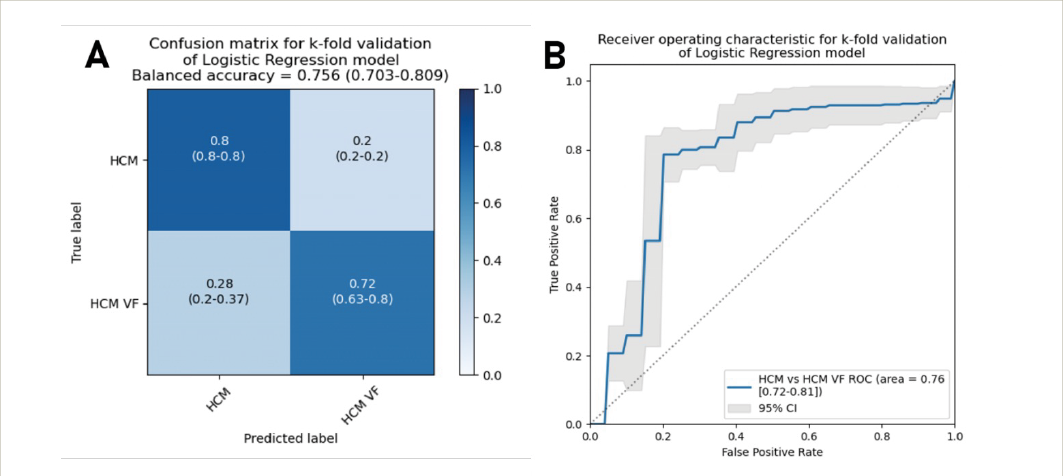
In contrast, following peak exercise, ECGi could demonstrate that the pooled HCM group had longer mean AT (60.1 ± 9.4 ms vs 52.2 ± 4.3 ms, p<0.001), activation dispersion (55.2 ± 16 ms vs 48.6 ± 12.1 ms, p=0.026) and mean ARI (227 ms vs 217 ms, p=0.016). These differences persisted into end recovery. HCM VF survivors could be differentiated from HCM patients without personal history of life-threatening arrhythmia. Following peak exercise, mean activation time (63.2 ± 7.2 ms vs 57.4 ± 10.3 ms, p=0.007), activation gradients (0.45 ± 0.11 ms/mm vs 0.36 ± 0.18 ms/mm, p=0.011) and ARI (234.0 ± 19.8 ms vs 221.4 ± 16.8 ms, p=0.026) were higher in HCM VF/VT. The differences in mean AT and ARI persisted into full recovery. Logistic regression was used to analyse electrical variables differentiating the HCM and HCM VF/VT groups. In k-folds validation, the model including mean AT and mean ARI produced an area under the receiver operating characteristic curve of 0.76 (95% confidence interval 0.72–0.81), with optimal sensitivity of 78.6% and specificity of 79.8% (Figure 1).
Conclusions: The HCM epicardial electrotype is characterized by slow, dispersed conduction and delayed, dispersed repolarization and activation-recovery intervals. ECGi is more sensitive to these differences than surface ECG. Combination of electrophysiological measures by logistic regression can improve differentiation over single variables. Future studies could test such models prospectively for risk stratification of sudden death in HCM.
Figure 1