Background: Common cut-offs for dense scar and ‘low-voltage areas’ on atrial 3D electroanatomical mapping are 0.05 mV and 0.50 mV, respectively. These are based upon statistical assessment of left atrial voltages in healthy individuals, rather than physiological assessment and may not fully represent underlying scar compared with unipolar readings or the novel parameter local impedance (LI).
Objectives: Investigate the relationship of bipolar voltage, unipolar voltage and LI with pacing threshold (PT). Establish cut-offs for electrically inert (EI), partially active (PA) and electrically active (EA) atrial tissue based upon pacing capture.
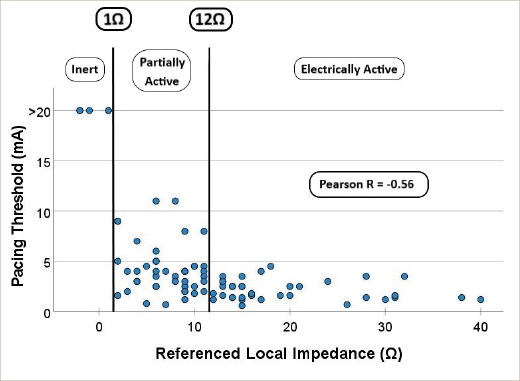
Methods: Patients undergoing AF ablation were recruited. Ultra-high-density 3D electroanatomical maps of both atria were created in Rhythmia HDx. PTs were assessed using the IntellaNav MiFi and Stablepoint (SP) ablation catheters between the tip and first ring electrodes. With the latter, a contact force of 10 g was targeted. PTs were assessed with a 2 ms pulse duration commencing at 5 mA and moved up or down accordingly. Sites without capture had PT recorded at the maximum tested 20 mA. LI was referenced against a blood pool reading (LIr) and analysed separately for MiFi and SP catheters. EI sites were defined as no pacing capture, PA capture between 5–20 mA and EA less than 5 mA. As no previous work has defined a PT to determine underlying atrial scar, a level was sought that would reflect EA tissue with consistently low PTs and PA tissue with significant variation in PTs. 5 mA was selected after review of data on scatterplots. Receiver operator and precision recall curves were plotted to define cut-offs.
Results: A total of 292 PTs and LI readings (202 MiFi, 90 SP) were recorded in 40 patients (MiFi: 20, SP: 20; male: 21/40; age: 66.7 ± 8.8 years; paroxysmal AF: 10, persistent AF: 30; de novo 21, redo 19). A weak correlation exists between SR voltages and PT (Pearson’s R – bipolar: -0.25, unipolar: -0.21, both p<0.0005). Weak correlations exist between LI and PT (Pearson’s R – MiFi = -0.35, SP = -0.33, both p<0.0005), but moderate–strong correlations exist between LIr and PT (R – MiFi = -0.52, SP = -0.56, both p<0.0005 [Figure 1]). For the EI–PA cut-off, the ability to distinguish capture/no capture for bipolar voltages was fair (AUROC = 0.74, optimal cut-off 0.03 mV), but poor for unipolar (AUROC = 0.68). In contrast, LIr had excellent discriminatory ability (AUROC – MiFi = 0.87, SP = 0.90, both optimal cut-offs 1Ω). For the PA–EA cut-off, the ability to distinguish PT above/below 5 mA for bipolar voltages was fair (AUROC = 0.69, optimal cut-off 1.86 mV), and poor for unipolar voltages (AUROC = 0.63). In contrast, LIr had good discrimination (AUROC – MiFi = 0.82, optimal cut-off 13Ω, SP = 0.89, optimal cut-off 12Ω). Using these cut-offs, there was a significant difference between PTs between EI, PA and EA, for bipolar voltage (EI: 20 mV, PA 6.0 ± 6.3 mV, EA: 2.6 ± 2.6 mV, p<0.0005) and LIr (MiFi: EI: 20 mV, PA 7.8 ± 7.5 mV, EA: 2.8 ± 2.8 mV. SP: EI 20 mV, PA: 4.0 ± 2.5 mV, EA: 2.2 ± 0.9 mV, both p<0.0005).
Conclusions: PTs suggest LIr is a superior marker in assessing electrical viability of atrial tissue over conventionally used voltage cut-offs. Based on pacing capture data, what has previously been labelled dense scar is not electrically inert and should be ablated if clinically indicated. Values of 0.03 mV and 1.86 mV, and for potential future LIr mapping, 1Ω and 13Ω are suggested as dense scar and low voltage area thresholds. ❑
Figure 1