Atrial fibrillation (AF) is the most common cardiac arrhythmia observed in clinical practice worldwide.1 The incidence of AF is around 0.1% in individuals under 55 years old and 9% in those aged over 80.2 The global prevalence of AF is estimated to be around 44 million, which is approximately 0.5% of the total world population.3,4 Therefore, AF represents a significant public healthcare burden. Moreover, several parameters can increase the susceptibility of AF, such as ageing, hypertension and diabetes mellitus (DM).5–7 In a meta-analysis conducted in 2018, patients with DM showed a 49% higher risk of AF compared with the general population, and with adjusting for the risk factors of hypertension, obesity and heart disease, the relative risk was 23%.8 In addition, the incidence of various adverse outcomes is markedly increased in patients with DM and AF, compared with the general population. In 2017, a cohort study from ORBIT-AF (Outcomes Registry for Better Informed Treatment of Atrial Fibrillation) showed that patients with AF and DM had a lower quality of life, and higher risk of both all-cause and cardiovascular mortality, sudden death, as well as cardiogenic and non-cardiac hospitalization compared with patients with AF free of DM.9,10
With an increasing burden of DM in recent years, its occurrence with comorbid AF represents a unique challenge. Patients with DM and AF have several adverse outcomes, but effective treatment strategies and prevention measures remain unclear. Rhythm control, along with restoration and maintenance of sinus rhythm are important for preventing cardiovascular complications. Previous animal studies have shown that DM can reduce the effects of anti-arrhythmic drugs, but there is currently a lack of definitive clinical trial data regarding their efficacy in humans.11 Catheter ablation is a non-pharmacological approach that can be applied in the setting of DM for treating AF that is refractory to medical therapy.7 As a procedure, it can significantly improve quality of life, with data supporting its superiority relative to anti-arrhythmic therapy for long-term prognostic outcomes.12,13
The purpose of this narrative review is to summarize the underlying mechanisms governing the pathogenesis of AF in DM, discuss the benefits of catheter ablation in treating patients with DM and AF, detail potential clinical predictors of AF recurrence following catheter ablation, and illustrate possible areas of further study of AF within the context of DM.
Pathogenesis of atrial fibrillation in diabetes mellitus
The pathogenesis of AF in DM is complex (Figure 1), involving an intricate interplay of electrical and mechanical factors that coordinate in tandem to induce arrhythmia. Many of the underlying mechanisms that contribute to the development of AF in type 1 (T1DM) and type 2 DM (T2DM) are similar, as these adverse pathways tend to be initiated by components common to both phenotypes, namely glucose intolerance, a pro-inflammatory environment and oxidative stress.14 Much of the available evidence suggests that these mechanisms primarily revolve around atrial structural and electrical remodelling, as well as autonomic dysfunction.14–16 From a macroscopic perspective, structural abnormalities typically initially manifest as subendocardial interstitial fibrosis, which is subsequently reflected at the organ level as gross chamber enlargement, including left atrial dilatation and ventricular hypertrophy.17,18 Such aberrations in myocardial structure adversely impact electrical function and enhance the risk of AF in patients with DM.19–23
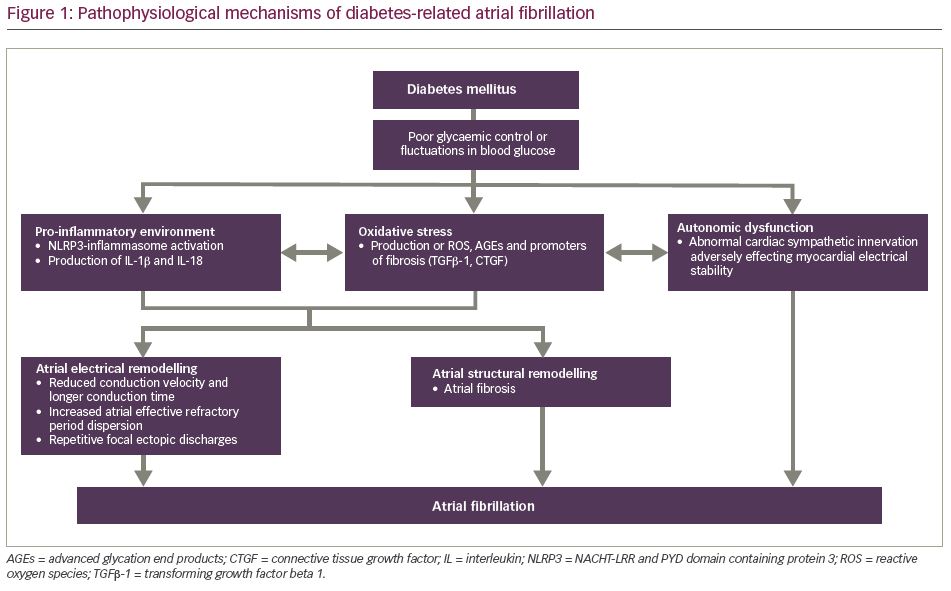
In one study comparing patients with and without DM, a high proportion of patients with DM had complex fractionated atrial electrograms (CFAEs) on more than 25% of their native atrial area, compared with patients without DM.24 CFAEs represent localized regions of the atria marked by reduced conduction velocity, as well as the presence of re-entry pivot points, both of which potentially stem from underlying fibrotic activity.25–27 Identification of CFAEs has implications for treatment, as they have shown to be promising targets for ablation. Current evidence indicates that pulmonary vein isolation in combination with CFAE ablation is superior in minimizing AF recurrence relative to either strategy used singularly.28 Many of the aberrations observed in diabetic hearts have also been studied on a microscopic level, providing insight into the many molecular pathways that work together to manifest the AF phenotype. The current school of thought is that the enhanced cardiac fibrosis and conduction disturbances that are typically observed in diabetic hearts chiefly occur secondary to many factors, including, but not limited to, inflammation and enhanced oxidative stress.29–32
Inflammation, oxidative stress and other factors in diabetes mellitus
First and foremost, inflammation has an important role in mediating cardiac fibrosis and subsequent electrical abnormalities in DM.29–32 There is much evidence to support the presence of an underlying chronic systemic inflammatory environment in patients with DM.33 Data from a recent rabbit model revealed enhanced activity of the NACHT-LRR and PYD domain containing protein 3 (NLRP3)-inflammasome in DM cardiomyocytes, which has been implicated in the development of both cardiovascular atherosclerotic disease and structural remodelling through left atrial fibrosis.34,35 Specifically, increased NLRP3-inflammasome activation in DM not only leads to the release of inflammatory cytokines that adversely affect the myocardium, namely interleukin (IL)-1β and IL-18, but also independently facilitates the creation of a re-entry substrate to maintain AF by shortening the atrial refractory period and inducing ectopic activity.34 Moreover, the importance of this pathway in AF development has also been showcased in studies focusing on the results of reduced NLRP3-inflammasome activity on the atria. These investigations lend further credence to the pathological role of diabetic inflammation in AF pathogenesis by demonstrating that inhibition of the NLRP3-inflammasome by glibenclamide modulates atrial structural remodelling, and reduces both conduction heterogeneity and overall AF inducibility.34
Another factor central to the development of atrial abnormalities implicated in AF is oxidative stress.29–32 DM induces upregulation of reactive oxygen species (ROS), which, in turn, stimulates the synthesis of collagen by cardiac fibroblasts, thereby leading to fibrosis.30,36–38 This ROS production is increased when patients with DM have either poor control of, or fluctuations in, blood glucose levels.39,40 The phenomenon of oxidative stress itself also facilitates the release of various atrial remodelling-related proteins that drive the progression of myocardial fibrosis, including transforming growth factor (TGF)β-1, a well-defined promoter of fibrosis that mediates cellular hypertrophy, extracellular matrix (ECM) synthesis and consequent structural remodelling.41–45 In addition to this, advanced glycation end products (AGEs) formed in the hyperglycaemic environment also serve as significant sources of ROS. AGEs binding to their receptors initiates signalling cascades that not only drive atrial fibrosis by enhancing the expression of connective tissue growth factor, but also aggravate myocardial inflammation and the subsequent development of diabetic cardiomyopathy by interacting with myeloid differentiation factor 2.46,47
In addition to the increased NLRP3-inflammasome activity and ROS production that serve as a cornerstone for the initiation, maintenance and subsequent progression of AF in DM, the existing theories also propose the role of various other factors in the pathogenesis of AF in DM.35 Particularly noteworthy, recent findings have illustrated that the diabetic environment activates the expression of human antigen R (HuR) in cardiac- and bone marrow-derived macrophages, which is an RNA binding protein that regulates the expression of many genes involved in inflammation, cell growth and fibrosis.48 This increased expression is accompanied by enhanced HuR nuclear-to-cytoplasmic translocation and exosomes transfer. These exosomes from macrophages, in the setting of hyperglycaemia, in turn drive cardiac fibrogenic reactions.46 Furthermore, recent investigations have also demonstrated the significance of atrial mitochondrial dysfunction in diabetic hearts.49–51 Rodent models have displayed an impairment in mitochondrial biogenesis, respiratory function and membrane depolarization in the setting of DM. These abnormalities were shown to be effectively counteracted with the use of sodium–glucose co-transporter-2 (SGLT2) inhibitor and dipeptidyl peptidase-4 (DPP-4) inhibitor drugs, which in turn may potentially have a use in the prevention of AF in DM.49–51
Atrial electrical disturbances in diabetes mellitus
Atrial electrical disturbances in diabetic hearts have been postulated as yet another factor that increases patient susceptibility to AF. Several investigations involving animal DM models have reported reductions in conduction velocity, lengthening of conduction time, increases in atrial effective refractory period dispersion and the existence of focal repetitive ectopic discharges, all of which collectively enhance the risk of AF.20,42,52,53 Research using insulin-resistant rats has shown an increase in intracellular Ca2+ stores within atrial cardiomyocytes, Ca2+ transient duration and the rate of sarcoplasmic Ca2+ sparks and leakage. Further evidence of dysregulation in Ca2+ handling has been supported by the abnormal upregulation of Ca2+ homeostasis-related proteins, namely Ca2+/calmodulin-dependent protein kinase II (CaMKII). Such aberrations in cardiomyocyte Ca2+ signalling can induce ectopic activity and AF.54 Moreover, data from a recent investigation proposed the presence of circulating immunoglobulin G autoantibodies within diabetic mice, which are said to in turn activate the PLC/IP3 pathway in atrial cardiomyocytes.52,55 These findings all but emphasize the central role of Ca2+ homeostatic dysregulation as a potential mechanism to explain the prevalence of AF amongst patients with DM.
It should be noted that electrical remodelling in diabetic hearts is not solely related to changes in Ca2+ signalling, but rather also to adverse alterations of cardiac ionic currents. Atrial cardiomyocytes of Zucker diabetic fatty rats have been shown to manifest with a decreased density of Kv4.3 channels and corresponding transient outward K+ currents (Ito), as well as decreased density of Kv1.5 channels and corresponding delayed rectifier K+ currents (IKur).20 These currents participate in atrial repolarization, and the consequent reduction in their expression can potentially precipitate AF.56,57 Particularly noteworthy, formerly discussed oxidative stress mechanisms can interact with and exaggerate electrical remodelling processes by promoting the downregulation of small conductance Ca2+-activated K+ (SK) channels in mice atria, serving as yet another substrate for arrhythmogenicity. Collectively, these ion channels are all closely related to the intrinsic electrical conduction system of the heart; as a result, subsequent disturbances in their ion transport capacities within the setting of DM likely work in a coordinated fashion to increase the risk of AF.
Furthermore, beyond the changes in transmembrane ionic transport, hyperglycaemia also modulates the expression of proteins involved in myocardial electromechanical coupling, including Connexin-43, a primary gap junction protein found through the atria and ventricles responsible for mediating cardiac conduction.58,59 Due to the central importance of such proteins to cardiac electrical homeostasis, any alterations in their synthesis or signalling induce electrical disturbances that enhance arrhythmic risk.60–62 Further evidence of such electromechanical dysfunction was shown in alloxan-induced diabetic models analysed by Doppler imaging. Relative to controls, left atrial lateral wall Pa’-start interval and right atrial wall Pa’-start were significantly higher in DM groups. Correlation analysis revealed an association between these parameters and subsequent cardiac fibrosis and delayed atrial electrical conduction times.63
Autonomic dysfunction in diabetes mellitus
The final area worthy of exploration relating to the pathophysiology of AF in DM is autonomic dysfunction. The general consensus regarding the role of neural remodelling in DM is somewhat unclear. Current evidence supposes that patients with DM have diminished cardiac sympathetic innervation that pathologically influences the electrical stability of the myocardium.64 More specifically, however, the diabetic phenotype is likely characterized by heterogenous sympathetic innervation, as adrenergic stimulation has been shown to enhance AF occurrence and atrial effective refractory period variability in rodent models.65 More in-depth analysis into the roles of neuronal remodelling within the setting of DM is required to fully comprehend the impact of the autonomic nervous system on AF development.
Catheter ablation
Catheter ablation has already been established as one of the effective therapies for patients with AF to restore sinus rhythm.66–68 According to the research of 521 centres from 42 countries in four continents, the efficacy of catheter ablation in the treatment of patients with either paroxysmal, persistent or long-lasting AF was about 80%, and approximately 70% of patients were not required to take antiarrhythmic drug therapy (ADT) during follow-up.69 Compared with ADT, catheter ablation has demonstrated more significant clinical benefits in patients with DM. A cohort study of 70 patients with T2DM in 2009 showed that the recurrence of AF and the hospitalization rate was higher in the ADT group, relative to that of catheter ablation group.70
Previous cohort studies on the efficacy and safety of catheter ablation for AF in DM have demonstrated conflicting results, as summarized in Table 1.70–79 The ablative approach adopted in patients with DM is largely the same as that which is taken in patients without diabetes or in those with other comorbidities. In a multicentre, prospective study from Germany, which enrolled 8,175 patients who underwent catheter ablation for AF or atrial flutter, 944 patients with DM were included. During the 1-year follow-up, there was no significant difference in arrhythmia recurrence between the patients with DM and patients without DM (46.4% versus 46.8%; p=0.90).72 In contrast, a study conducted by Chao et al. showed a higher arrhythmia recurrence rate in patients with abnormal glucose metabolism.71 This discrepancy can potentially be justified by the differences in baseline characteristics of the subjects included, whereby patients without diabetes appeared to present with a high incidence of comorbidities, including structural and coronary heart diseases, that could have possibly accounted for the similar rates of AF recurrence observed between them and their DM counterparts.72
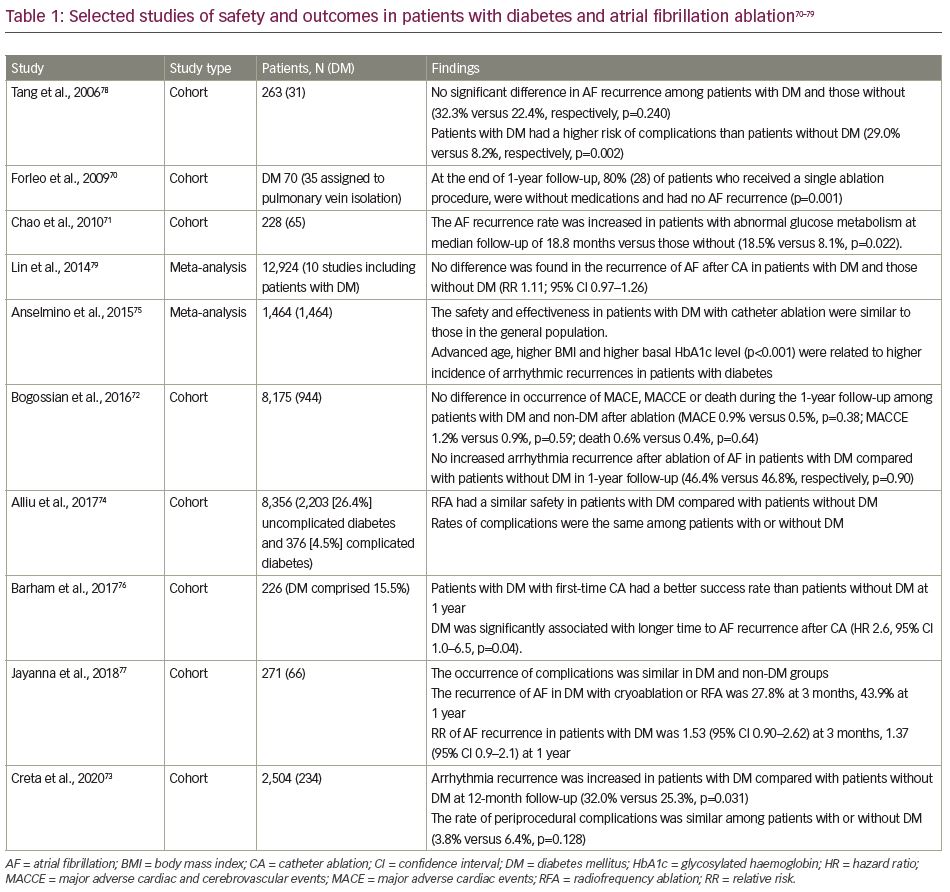
Another investigation by Creta et al. similarly demonstrated a comparatively higher rate of AF recurrence in patients with DM than in patients without DM.73 This study performed propensity-matching in order to circumvent the limitation of disparities in baseline characteristics between subjects. As such, these findings potentially support the role of aberrations in glucose tolerance in the pathophysiology of AF, likely through more severe alterations in atrial remodelling and conduction abnormalities in subjects with DM.66,68 In an attempt to shed some light on the differences in AF incidence post-ablation demonstrated in different observational investigations, a systematic review was performed in 2015. In this meta-analysis of 1,464 patients with a follow-up of 27 months, efficacy in maintaining sinus rhythm was similar between DM and non-DM groups.80 Their meta-regression analyses demonstrated that increasing age, higher body mass index and higher baseline glycated haemoglobin (HbA1c) were associated with higher recurrence rates. These findings would suggest that catheter ablation can be effective in younger patients with DM with good glycaemic control.
Regarding procedural safety, Tang et al. reported that the rate of complications following catheter ablation for AF, such as haematomas, cardiac tamponade and stroke, was higher in subjects with DM than those without.78 However, in another larger cohort study, there was no statistically significant difference in the odds of complications for patients with DM compared to patients without DM.74 Overall, despite the findings of some studies of complications and poor outcomes in the context of DM after catheter ablation, including but not limited to stroke, thrombosis, haematoma and cardiac tamponade, the majority of the investigations examining safety outcomes of the procedure in AF treatment have not displayed any significant differences between subjects with and without DM.71,78
Predictors of atrial fibrillation recurrence following catheter ablation in diabetes mellitus
The electrocardiogram (ECG) is a convenient and non-invasive diagnostic test and risk predictor. In a general population, corrected QT (QTc) interval has previously been shown to be predictive of incident AF.81 Specifically, for patients with DM undergoing catheter ablation for AF, a 2016 study (mean follow-up: 29.1 ± 12.4 months) reported longer QTc intervals in those with AF recurrence compared with those without (425.2 ± 21.5 ms versus 414.1 ± 13.4 ms, p=0.002). For every 10 ms prolongation in QTc, there was a 29% higher risk of AF recurrence (hazard ratio 1.286, 95% confidence interval 1.039–1.591, p=0.021). A cut-off of 418 ms showed a sensitivity of 55.7% and specificity of 69.9% for classification (area under the curve 0.65).82
Several alternative ECG parameters have also been investigated in relation to AF recurrence post-ablation in AF cohorts, albeit a certain proportion of patients did in fact present with baseline DM. Amongst assessed parameters, it has been shown that prolongation of PR interval, as well as smaller P wave duration variations in leads II and III, which refer to the difference between post- and pre-ablation P wave duration, are significantly associated with AF recurrence following multivariate adjustment.83,84 Moreover, there is also an abundance of evidence to support a correlation between ECG atrial or ventricular repolarization parameters, such as QTc dispersion, P wave dispersion and Tpeak-Tend interval/QTc ratio, with worsening hyperglycaemia in patients with DM.85 Overall, further investigation is still required to examine the use of P wave duration and PR interval, as well as potential atrial and ventricular repolarization parameters, to predict AF recurrence because of their lack of specificity in patients with DM. Nonetheless, despite existing uncertainties, preliminary data from the studies above showcase the strong potential of ECG methods to predict AF recurrence after ablation in patients with DM within clinical settings.
Beyond ECG parameters, efficacy of blood glucose control can also influence the recurrence rate of AF after ablation. Control of glycaemic levels is crucial for DM management, and is often heterogenous within patient populations. In a notable study of 420 patients with DM who underwent AF ablation, subjects in the high fasting blood sugar group (>110 mg/dL) reportedly had an increased risk of AF recurrence compared with those with normal fasting blood sugar, thereby emphasizing the importance of adequate glycaemic control for DM prognosis.86 Another investigation, focusing instead on HbA1c levels, indicated that an elevated HbA1c was predictive of post-ablation AF incidence. Specifically, a value of HbA1c ≥6.9% presented with a sensitivity of 55.0% and specificity of 67.4% for AF risk prediction.87 Moreover, there are also data to suggest that patients with a worsening trend of HbA1c 1 year prior to ablation are more likely to develop AF following the procedure relative to patients who experienced improvements in glycaemic control during the same time period, in turn indicating that a period of optimal glucose control before ablation may have an impact on prognosis.88
Collectively, the findings of the aforementioned investigations suggest the prospective use of blood glucose control efficacy as a potential factor to assess arrhythmogenicity following catheter ablation. However, it should be noted that a possible limitation in the implementation of this parameter resides primarily in its lack of specificity to AF, as worsening glycaemic control has, unsurprisingly, demonstrated an association with a wide range of non-arrhythmic cardiovascular, cerebrovascular and renal outcomes as well.89
In tandem with glycaemic control, the implementation of echocardiographic parameters to examine the risk of AF recurrence after ablation has also been assessed. In a cohort of 149 patients with T2DM, left atrium size ≥39.0 mm predicted AF recurrence with 70.0% sensitivity and 55.1% specificity, even after multivariate adjustment for HbA1c status and other parameters.88 Although there is a relative paucity in literature assessing the use of echocardiographic parameters to predict AF recurrence following ablation in patients with DM, many other cohort studies have illustrated the use of other variables to predict post-procedural freedom from AF, including, but not limited to, the ratio of mitral peak velocity of early filling (E) to early diastolic mitral annular velocity (E’) (E/E’ ratio) and left atrial appendage flow velocity.90 These findings, in turn, warrant further investigation into the use of these variables as possible markers of AF recurrence, specifically in diabetes cohorts.
Potential areas for further study regarding the development of atrial fibrillation in diabetes mellitus
Despite the abundance of literature pertaining to the relationship between DM, glycaemic control and the development of AF, there are several domains of this topic that warrant more investigation.91 The notion of DM as an independent predictor of AF has long since been established, and given the aforementioned mechanisms through which this relationship comes to fruition, it is not surprising to find data proposing a positive correlation between both poor glycaemic control and DM duration with AF development.92 However, there lacks sufficient research into the link between these two variables and the age of onset of AF. From a mechanistic perspective, a longer DM duration and poorer glycaemic control presumably translate into greater hyperglycaemic-induced oxidative stress and inflammatory cytokine production, which intuitively may lead to a faster rate of or more severe atrial structural and electrical remodelling that predisposes to earlier AF development.93 Although this notion may seem sound in theory, further investigation is still needed to determine the precise timing of AF onset in DM in order to establish its validity, along with its prospective influence on disease monitoring and treatment in the clinical setting.
It may also be worth assessing the likelihood of AF reversion in subjects with DM with optimal glucose control. Weight gain or obesity has been shown to be associated with suboptimal glycaemic control and poorer glucose tolerance.94,95 Data from a recent animal model has showcased that a reduction in weight is associated with a corresponding decline in inflammation, atrial fibrosis and conduction heterogeneity, leading to a reversal of atrial remodelling and AF inducibility.96 Nonetheless, whether weight reduction and, by extension, optimal glycaemic control, can themselves reduce the recurrence of AF in patients is as of yet uncertain, and, therefore, requires further study.
It is also abundantly clear that there is much unknown pertaining to the relative efficacy of different anti-hyperglycaemic drugs in the prevention of AF. A study examining the effects of insulin, metformin, sulfonylureas, thiazolidinediones and DPP-4 inhibitors, demonstrated that although patients on insulin were more prone to AF development, those on metformin and thiazolidinediones showed a reduction in AF incidence, whereas patients on sulfonylureas and DPP-4 inhibitors experienced no change in AF risk.97 However, despite evidence to support these findings, many other studies have not maintained this consistency.98–103 Regarding other classes of anti-hyperglycaemic drugs, glucagon-like peptide-1 receptor agonist users have also presented with varying results, as AF risk has reported to be increased with albiglutide, but unchanged with semaglutide, liraglutide and dulaglutide.104,105 In contrast, SGLT2 inhibitors have recently showed efficacy in AF prevention.106
With the contradictory findings concerning AF development associated with medical therapy, further study is still required in order to determine which of the available classes of anti-hyperglycaemic drugs is the most suitable option. In tandem with this, investigations have also been conducted to examine the role of such drugs in the context of catheter ablation. In a cohort of patients with DM with drug-refractory paroxysmal AF, treatment with pioglitazone prior to ablative therapy, significantly reduced the risk of AF recurrence.101 Similarly, the use of metformin before catheter ablation in a group of subjects with DM and AF also reportedly reduced the risk of recurrent arrhythmias.107 Despite the clear effectiveness of anti-hyperglycaemic medications in modulating disease recurrence following catheter ablation, the mechanisms through which such pharmacotherapy can prevent AF – be it directly through glycaemic control or indirectly through some of their anti-inflammatory and anti-oxidative properties – is still unknown and necessitates greater study.
Furthermore, another pertinent point of contention in existing literature is the precise nature of the interplay between DM, AF and incident stroke. Data from a large set of subjects with comorbid DM and AF revealed a significant association between a per unit increase in HbA1c levels and incident stroke after adjustment for CHA2DS2-VASc score risk factors.108 These findings are supported by those obtained from a large Danish registry, which revealed a higher risk of ischaemic stroke in patients with HbA1c values of 49–58 mmol/mol and >58 mmol/mol, compared with those with values ≤48 mmol/mol. However, it should be noted that, although this relationship remained in patients with DM for <10 years, it was surprisingly not maintained in patients with DM ≥10 years, wherein higher HbA1c values failed to show any correlation with stroke risk.109
The importance of DM duration was further highlighted in the ATRIA California community-based cohort study, in which DM duration ≥3 years was associated with an increased incidence of ischaemic stroke relative to DM duration of <3 years.110 Contrary to these studies, there is also some evidence to suggest an absence of any enhanced thromboembolic risk amongst patients with AF.111 Moreover, it is also necessary to contextualize such findings in relation to anticoagulant therapy for stroke prevention.
Despite current guidelines, an underuse of anticoagulant therapy in patients with AF remains a persistent dilemma in healthcare. Perhaps the most important component of anticoagulant management that needs to be considered during its administration is the maintenance of a balance between preventing bleeding and thromboembolism.112 In a group derived from another nationwide Danish registry, it was revealed that longer DM duration was continuously related to a higher risk of thromboembolic events, but not to bleeding risk secondary to anticoagulant therapy, thereby possibly justifying an initiation of anticoagulation in patients with AF with prolonged DM.112 All in all, the varying reports from available investigations should incite the performance of more studies that can further clarify the precise role of glycaemic control and DM duration in stroke development secondary to AF, in order to more effectively guide the use of anticoagulant therapy in the clinical setting.
Conclusions and perspectives
In conclusion, both DM and AF have individually and collectively contributed to a significant burden on healthcare systems and economies worldwide. AF has been proven to manifest with a higher incidence in DM cohorts, and has in turn garnered greater, much warranted attention as to its pathophysiology in the setting of hyperglycaemia. The underlying mechanisms governing AF initiation and progression in DM populations is still somewhat uncertain, with factors pertaining to systemic inflammation, oxidative stress and ion channel dysfunction likely collaborating and interacting co-ordinately to adversely remodel the diabetic heart.
Catheter ablation is a widely implemented treatment method used for the recovery of sinus rhythm in patients with AF, including AF with DM. Whilst most studies fail to show any differences in post-procedural complication rate, there is some evidence to suggest a higher incidence of post-procedural complications amongst subjects with DM than those without DM. This, coupled with the lack of clearly defined variables, such as ECG parameters or glycaemic status, to assess for patient prognosis post-ablation, poses an additional problem towards clinical management. As such, more studies are still required to not only further elucidate the mechanism of AF susceptibility in DM, as well as the corresponding optimal therapeutic strategies for these patients, but also to provide insight on potential predictors of AF recurrence in patients with DM, particularly in relation to indices that can be obtained easily, efficiently and non-invasively.







