Introduction: The Micra post-approval registry (PAR) and Micra Acute Performance regional cohort (MAP; conducted in Europe, Middle East, and Africa [EMEA]) are ongoing prospective registries designed to evaluate safety and performance of the Micra transcatheter pacing system in the post-market real-world setting. The patient profile and implant characteristics of patients undergoing Micra implant in the UK were compared with the rest of the EMEA region.
Methods: Patients undergoing Micra implant attempt and enrolled in the Micra PAR or MAP cohorts in the EMEA region were included in the analysis. Baseline characteristics and procedural outcomes were compared between those patients in the UK and the rest of the EMEA region.
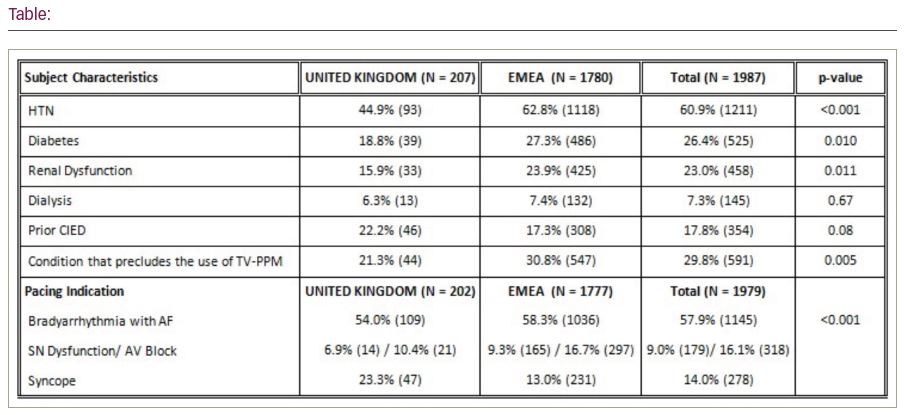
Results: In the UK a total of 207 of 1,987 (10.4%) patients underwent Micra implant attempt at 13 centers between September 2015 and January 2020. The Micra device was successfully implanted in 206 of the 207 patients (99.5%), similar to the 99.6% implant success rate in the EMEA region. There were comparable sex distributions with males 68.6% (142) in the UK and 62.2% (1,170) in the EMEA. Patients in the UK tended to be significantly younger than patients from other EMEA countries (68.3 ± 17.8 vs 76.7 ± 12.8, p<0.001) and age was no bar to implantation with similar ranges (18–95 vs 13–102). UK patients were less likely to have a history of hypertension, diabetes, or renal dysfunction (all p<0.05; Table). Similar to the rest of EMEA, the most common primary pacing indication in the UK was bradyarrhythmia with atrial fibrillation; however, syncope was a more common primary pacing indication in the UK (23.3% vs 13.0%). The majority of implants in UK patients (92.8%) required ≤3 deployments (EMEA 92.1%). Average pacing thresholds among UK patients at implant were 0.6 ± 0.4 V at 0.24 ms, comparable to the EMEA pacing thresholds at implant (0.6 ± 0.5 V at 0.24 ms).
Conclusion: Micra patients from the UK were younger, had fewer comorbidities, and differing primary pacing indications than those of patients from the rest of EMEA. Importantly, the Micra transcatheter pacemaker was implanted with a high rate of success among patients from both the UK and the rest of EMEA despite these differences in patient baseline characteristics.