Introduction: Subcutaneous ICD (S-ICD) was designed to avoid complications associated with transvenous ICDs. But not all patients are eligible for S-ICDs and eligibility is identified after screening S-ICD candidates using guidelines by the device manufacturer. The S-ICD is particularly valuable for younger patients such as congenital heart disease (CHD) and hypertrophic cardiomyopathy (HCM) patients requiring longer defibrillation protection, but information regarding suitability for S-ICD in these populations is scarce with few studies reporting ineligibility rates between 24.6-56 % and between 7-38% for the CHD and HCM patients, respectively. These rates may be due to abnormal T-wave morphology resulting from structural changes that characterizes these patients’ groups. We report our experience with S-ICD screening in real-world population as a tertiary referral centre for complex cardiac devices in the UK.
Methodology: We performed a retrograde analysis on patients with an indication for an ICD who had screening for S-ICD eligibility at our centre between 2014 and 2021. Patients were considered eligible if they had at least one vector that passed the screening in two postures as recommended by the device manufacturer.
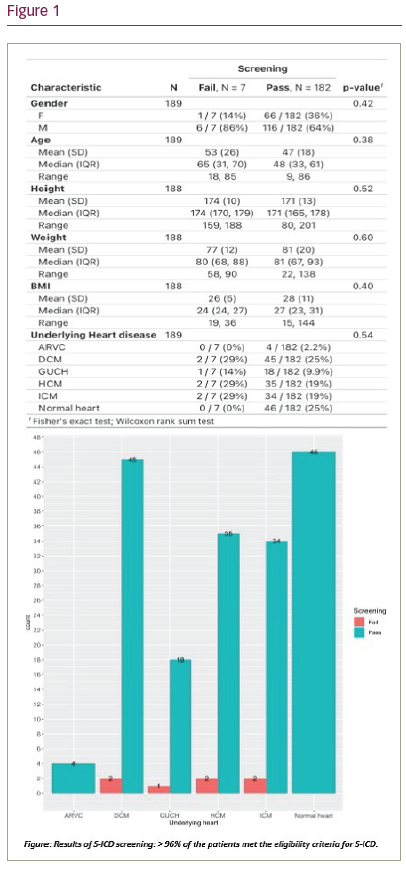
Results: A total of 189 (Mean age 47.2 years, 64.6% males) patients were included. Mean BMI was 27.9. Underlying heart conditions were dilated cardiomyopathy 47 (24.9%), structurally normal heart 46 (24.3%), HCM 37 (19.6%), ischaemic cardiomyopathy 36 (19%), CHD 19 (10.1%) and arrhythmogenic right ventricular cardiomyopathy 4 (2.1%). 182 (96.3%) patients met the eligibility criteria for an S-ICD. There was no statistically significant difference in the S-ICD eligibility based on age, sex, or BMI or between patients with different underlying heart conditions (Figure 1).
Conclusions: Our analysis showed that most of the patients- regardless of underlying cardiac condition- are likely to be deemed S-ICD eligible following current screening practices.