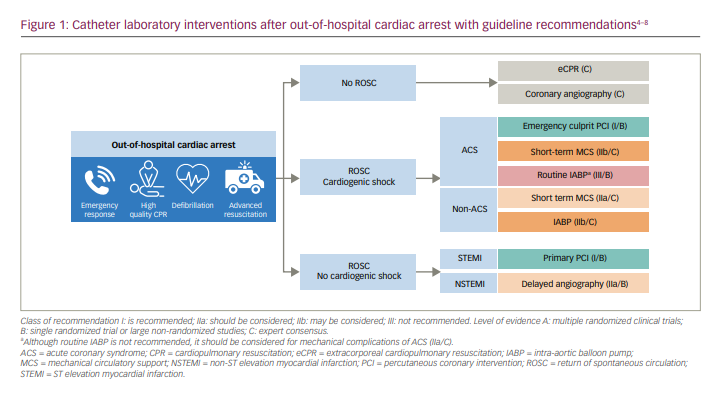
The true epidemiology of out-of-hospital cardiac arrest (OHCA) is difficult to define, with wide variations in reporting, management and outcomes worldwide.1,2 In Europe, the incidence of OHCA is estimated to be between 67 and 170 per 100,000 people and it continues to be a major public health challenge that is reliant on multiple links in a ‘chain of survival’ to improve outcomes.3 For patients who survive and are admitted to hospital, ongoing management is based upon organ support and the diagnosis and treatment of an underlying cause. This article focuses on recent developments that are changing our understanding about the role of the cardiac catheter laboratory in the resuscitated OHCA patient without ST elevation or cardiogenic shock (the third pathway depicted in Figure 1).4–8
The cardiac catheter laboratory
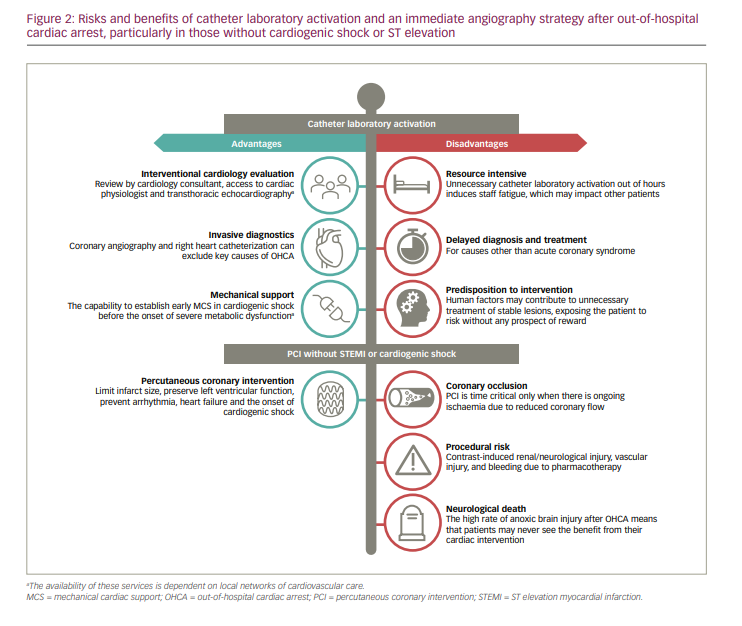
Activation of the cardiac catheter laboratory and its associated multidisciplinary team can provide a multifaceted service to the OHCA patient, the majority of which will have a cardiac cause for their arrest.9 First, it provides immediate diagnostic evaluation by the interventional cardiology team. The alternative strategy, for the patient to be taken to the nearest hospital regardless of primary capability in percutaneous coronary intervention (PCI), could result in heterogeneity of care due to regional differences, particularly with non-specialist hospitals. Second, invasive procedures such as coronary angiography and right heart catheterization may inform diagnosis and ongoing patient care, even when PCI is not indicated. Third, it provides the option for placement of percutaneous mechanical support devices for those in cardiogenic shock (Figure 1).
These benefits do have fiscal implications, however, and can potentially increase the risk to the patient (Figure 2). Provision of a 24-hour cardiac catheter laboratory is resource intensive, and out of hours, it may utilize staff that are required to work the following day. Coronary angiography can lead to delays in neuroprotective measures or in the diagnosis and treatment of non-coronary causes for cardiac arrest.9 Although coronary disease (either atherosclerotic due to plaque disruption, or non-atherosclerotic, such as spontaneous coronary artery dissection) is the predominant aetiology for cardiac OHCA, other causes such as cardiomyopathy, valvular heart disease, congenital heart disease and primary electrical abnormalities must be considered. It also places patients at risk for: 1) procedural complications, such as bleeding due to the use of pharmacological adjuncts; 2) renal injury due to the administration of iodinated contrast to kidneys that have already suffered an ischaemic insult;10 and 3) potential neurological injury from contrast crossing a compromised blood–brain barrier,11 coupled with any haemodynamic instability that may be induced by PCI.
This risk–benefit ratio is shifted in favour of catheter laboratory activation, however, when the probability of identifying clinically relevant coronary artery disease is high. In two observational studies, obstructive coronary disease (defined as >50% stenosis in these studies) was identified in 89–96% of those with ST elevation and in 51–58% in those without.12,13 However, perhaps more relevant than the mere prevalence of coronary disease is thrombotic coronary occlusion. For those with stable coronary disease, a considered assessment of myocardial ischaemia and viability may be helpful before embarking on revascularization.14 In thrombotic occlusion, intervention is time critical. In these cases, resolving cardiac ischaemia may lead to preservation of left ventricular function, improvement in haemodynamics and the prevention of reinfarction and cardiac death.15 Acute coronary occlusion has been reported to be present in over 80% of those with ST elevation. For those who have an initial shockable rhythm (ventricular fibrillation [VF] or ventricular tachycardia [VT]) without ST elevation, acute occlusion is reported in 23%.16 The presence of refractory VF or VT (persistent after three shocks delivered by emergency medical services and administration of amiodarone) increases this further to 64%.17
In cardiogenic shock, the prompt revascularization or exclusion of a coronary culprit is also essential, given that acute coronary syndromes (ACS) are responsible for 81% of cases and cardiogenic shock carries a devastating short-term mortality.18
However, even in the presence of ST elevation, electrical instability or cardiogenic shock, another layer of complexity is introduced after OHCA. Although conscious patients have a favourable prognosis and their outcome is similar to the non-arrested ACS group,12,19 after return of spontaneous circulation (ROSC) the majority will be comatose. Predicting the likelihood of neurological recovery in these patients is notoriously difficult.20,21 Therefore, while patients with ST elevation myocardial infarction (STEMI) or cardiogenic shock still undergo immediate angiography given its established benefits,18,22 the urgency with which patients without these features undergo coronary angiography is a matter of debate. Immediate catheter laboratory activation in these cases has the potential to expend significant resources, all within the initial 72 hours of uncertainty before any formal neuro-prognostication can be carried out.23
Current guidelines and practice
Wide variation exists in the management and outcomes of OHCA, including the use of coronary angiography after cardiac arrest, which reflects the lack of consensus and randomized data in previous years. In the UK national database of all catheter laboratory procedures, the rate of PCI undertaken in emergency ventilated patients as a proportion of total PCI within a centre (the majority of which are OHCA, and whose outcomes are censored from PCI operators’ publicly available data) varies widely, from 0.0 to 4.7%. This was independent of primary PCI volume or the provision of 24-hour PCI at that centre.24 This variation has been similarly demonstrated in the USA and in France.25–27 This is despite contemporary guidance at the time of these studies recommending angiography within <2 hours following cardiac arrest,28,29 supported by multiple meta-analyses of observational data.30–32
The European Society of Cardiology now recommends that delayed angiography should be considered for haemodynamically stable patients after cardiac arrest without STEMI (Table 1).5,33 This is consistent with the Society for Cardiovascular Angiography and Interventions consensus statement, which emphasizes that the OHCA group is a highly heterogeneous population and an individualized approach is required,33 with an assessment made of the factors that are likely to favour a good neurological recovery before proceeding to catheter laboratory activation. This change in recommendation reflects the publication of results from the first in a series of trials that have addressed the timing of angiography after cardiac arrest.34–36

A new era of randomized controlled trials in cardiac arrest
COACT
The Coronary angiography after cardiac arrest trial (COACT; Netherlands Trial Register: NTR4973) was an investigator-initiated, multicentre trial in Holland that assessed the effect of an immediate versus a delayed angiography strategy on 90-day survival in patients who remained unconscious after OHCA (VF/VT arrest), but had no ST elevation on their electrocardiogram (ECG).34,35 Angiography in the delayed arm was performed after neurological recovery, which was in general after discharge from the intensive care unit (ICU). Cardiogenic shock, refractory ventricular arrhythmia and a clear non-coronary cause were among the exclusion criteria. The trial was designed to detect a 40% difference in the primary outcome with 85% power, based on a predicted 45% survival in the immediate angiography arm and 32% in the delayed arm.
A total of 552 patients were randomized 1:1 from 2015 to 2018, with follow-up completed for 98%. There was no significant difference in the primary outcome of survival at 90 days: 64.5% in the immediate group versus 67.2% in the delayed group (odds ratio 0.89, 95% confidence interval [CI], 0.62–1.27). This was much higher than anticipated in the power calculation for the trial, and double what was predicted for the delayed angiography arm, indicating that this is a lower-risk cohort than the investigators predicted. Neurological causes were responsible for the majority (64%) of deaths.
Despite the established reduction in mortality and vascular complications with radial access (and the exclusion of cardiogenic shock making the need for mechanical support devices and femoral access unlikely),37 radial access was used in only 41% of the immediate angiography group versus 73% in the delayed group. Furthermore, there was increased use of aspirin (87% versus 76%) and P2Y12 inhibitors (71% versus 58%) in the delayed angiography arm. Both these factors may have disadvantaged the immediate angiography group and potentially diluted any beneficial treatment effects.
Commentary
A proportion of the COACT patient population can be expected to have coronary occlusion despite the absence of ST elevation, and this group perhaps offers the greatest opportunity for benefit from PCI. This was present in only 5% of those who underwent angiography, with a further 15% having an acutely unstable lesion, defined as a ≥70% stenosis with angiographic irregularity, dissection, haziness or thrombus. This is a small proportion of patients in whom an immediate PCI strategy could influence outcomes, let alone a 40% change in 90-day survival; hence, the trial is underpowered to assess benefit in this specific group.
Furthermore, the PCI rate in the immediate angiography arm was markedly higher than the rate of angiographic occlusion or unstable lesions (33% versus 17%). Defining an unstable lesion can be very difficult in non-STEMI. When cardiac magnetic resonance and late gadolinium enhancement are used to localise infarcted myocardium in the non-arrested population, an apparent angiographic culprit vessel matches the infarcted territory in less than half of patients.38 Nonetheless, this disparity suggests the revascularization of some stable lesions, which might expose the patient to the risks of PCI without any potential benefit. From the human factor perspective, we must resist the path of least resistance, which is for the interventional cardiologist to deliver a patient to intensive care ‘fully revascularized’ with no further intervention required, even if this is mutually beneficial for the intensivist, who then does not have to consider a return to the catheter laboratory as part of the patient’s treatment course. We should work with our colleagues to emphasize that the presence of coronary disease does not imply a cause for OHCA, nor is it alone a clear mandate for coronary intervention. The act of performing coronary angiography in itself (whether immediately or delayed) must not predispose the patient to unnecessary intervention.
Targeted temperature management (TTM) is the only neuroprotective intervention currently recommended after OHCA.23 The time to establish TTM (therapeutic hypothermia at 33–36°C at the time of these trials) was longer in the immediate angiography group (geometric mean of 6.5 [5.9–7.1] versus 5.5 [5.0–6.0] hours, CI 95%).39 However, the large and well-designed TTM2 trial (with more participants than all previous randomized studies in therapeutic hypothermia combined) recently showed no difference in mortality or functional outcome between normothermia and hypothermia at 33°C.40 It may be that hypothermia is simply a blunt tool for the avoidance of fever, which is known to be associated with poor outcomes. It therefore seems unlikely that the difference in time to TTM had a significant effect on outcomes in COACT.
COACT summary
In comatose patients after resuscitated VF or VT arrest and without ST elevation, a strategy of immediate versus delayed angiography did not show a significant difference in 90-day survival. A smaller effect size than 40% cannot be excluded, but any improvement in cardiac outcomes may also be concealed by the high proportion of deaths due to neurological injury. The lower-than-expected number of coronary occlusions may reflect patient selection, and limit extrapolation to an all-comers population.
TOMAHAWK
TOMAHAWK (Immediate unselected coronary angiography versus delayed triage in survivors of out-of-hospital cardiac arrest without ST-segment elevation; ClinicalTrials.gov identifier: NCT02750462) was an investigator-initiated, international, multicentre trial in Europe that assessed the effect of an immediate angiography versus a strategy of initial ICU evaluation on 30-day mortality in patients who had been resuscitated from OHCA and had no ST elevation.36 The delayed group could proceed to angiography after a minimum of 24 hours, or sooner if there was electrical instability, cardiogenic shock, new ST elevation, or troponin elevation to >70 times the upper limit of normal at 6 hours. Cardiogenic shock, electrical instability and a clear non-cardiac cause were among the exclusion criteria. The study was designed to reject the null hypothesis of no difference between groups with 80% power, based on previous observational data that predicted 34% mortality in those undergoing immediate angiography and 46% mortality with delayed angiography.
A total of 554 patients were randomized 1:1 from 2016 to 2019, with follow-up completed for 96%. There was no significant difference in the primary outcome of 30-day mortality: 54% in the immediate group versus 46% in the delayed group (hazard ratio 1.28, 95% CI 1.00–1.63). The prespecified secondary endpoint of all-cause death or severe neurological deficit occurred in 64.3% of the immediate group versus 55.6% in the delayed group (relative risk 1.16, 95% CI 1.00–1.34), which should be viewed as hypothesis generating.
A culprit lesion was identified in 40% of patients in TOMAHAWK, with a similar rate of PCI. However, the definition of a clinically relevant lesion warranting revascularization was left to the discretion of the operator. The rate of thrombotic coronary occlusion was not provided in this paper. Neurological injury was responsible for death in 40% of the patients.
Commentary and comparisons with COACT
It is worth highlighting some of the similarities and differences between COACT and TOMAHAWK (Figure 3).34–36 Due to difficulties in enrolment, the prematurely terminated PEARL (Early coronary angiography versus delayed coronary angiography; ClinicalTrials.gov identifier: NCT02387398) trial, which was conducted at the same time as these studies, has not been included for further comparison.41 The design of TOMAHAWK allowed for earlier coronary angiography in the delayed group, at a median of 47 hours (interquartile range [IQR] 26.1–116.6), versus 122 hours (IQR 52.0–197.3) in COACT where this was delayed until neurological recovery. A pragmatic approach allowed crossover from the delayed arm to angiography for set criteria suggestive of a coronary culprit. More extensive coronary disease was seen in TOMAHAWK, with 37% having three-vessel disease (versus 17% in COACT), although the rates of coronary artery bypass graft, PCI and previous myocardial infarction were similar. Common findings were the low rates of radial angiography and the high rate of neurological death (also 40%).
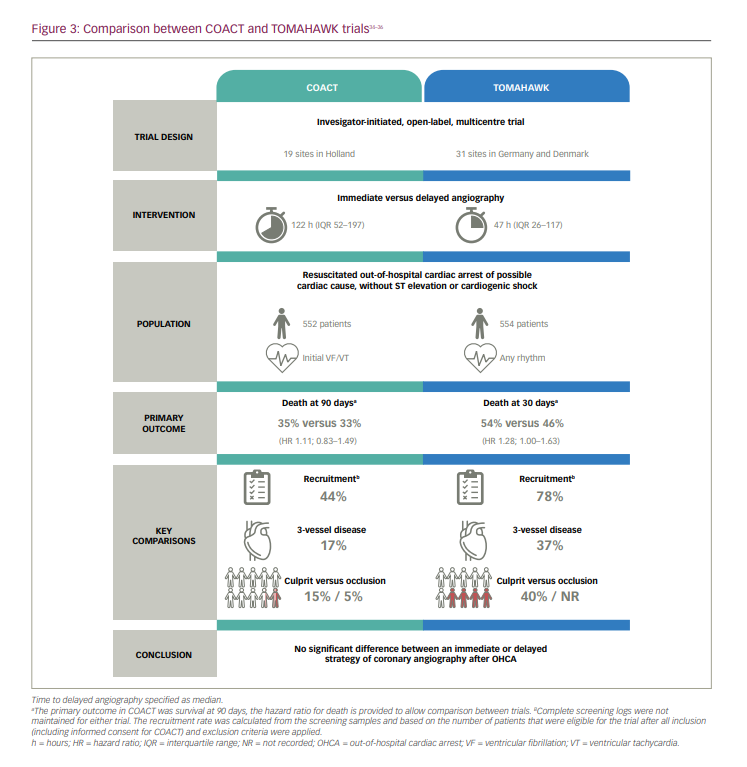
Do the reported numbers of acute coronary lesions in either of these trials represent the real-world prevalence of unstable coronary lesions after resuscitated and comatose VF/VT arrest? Observational series of patients without ST elevation would suggest identification of a culprit lesion in up to 33%, with vessel occlusion in up to 23% of patients.12,15,42,43 How do we account for this discrepancy? After satisfying all inclusion and exclusion criteria (including the provision of informed consent), over half of the patients included in the COACT screening sample were still excluded for unspecified reasons. Without the benefit of a COACT registry to track the outcomes of these patients, and the TOMAHAWK registry being abandoned in the final protocol, we do not know the true prevalence of coronary occlusion in either population. We can speculate that clinicians would have been unwilling to randomize patients with a high probability of a coronary culprit lesion, such as a history of chest pain prior to cardiac arrest or a markedly ischaemic ECG. It has been the established standard of care for several years to offer immediate coronary angiography ‘analogous to STEMI’ in those with dynamic ST-T-wave changes even in the absence of ST elevation.28 The proportion of patients that were excluded in TOMAHAWK despite being otherwise suitable was lower (22%), and it is possible that a more representative population was enrolled in this study.
Treating the heart versus protecting the brain
The COACT and TOMAHAWK trials confirm that many comatose patients after OHCA will ultimately go on to suffer neurological death. This poses a challenge when trying to ascertain the effects of an intervention such as immediate cardiac catheterization, as any longer-term benefits in left ventricular function, rehospitalization, reinfarction or cardiac mortality may be concealed by early neurological death. Even if these benefits do exist, it is clearly unhelpful to pursue them if the neurological outcome is futile.
When considering the role of the catheter laboratory in these patients, we should therefore focus on two priorities. First, how to select patients with a low risk of neurological death who are likely to see the benefits from any cardiac intervention. Second, how to select patients in whom the diagnosis or treatment of a coronary culprit should not be delayed. This is presumed in those with ST elevation, cardiogenic shock and electrical instability,5 but for patients with none of these features, can we identify those with a coronary occlusion who may benefit from immediate PCI?
Unfortunately, on the patient’s arrival, the clinician is often faced with the need to answer these questions based on limited and imperfect information. A patient’s history, comorbidities and wishes may be unknown. Therefore, decisions rest primarily on the characteristics of the resuscitation, the 12-lead ECG and results from point-of-care whole blood analysis.
Neurological recovery
The difficulty in predicting a patient’s likely neurological outcome at admission has led to the development of several risk scores (Table 2).44–48 The Cardiac Arrest Hospital Prognosis (CAHP) score is applied on hospital admission, and the Out of Hospital Cardiac Arrest (OHCA) score and Target Temperature Management (TTM) score are used at the time of ICU admission.44–46 They all perform reasonably well within their corresponding derivation cohorts in predicting neurological outcomes. In a large registry, early invasive angiography was independently associated with survival only in those with a low (<150) CAHP score (i.e. a presumed good outcome).27
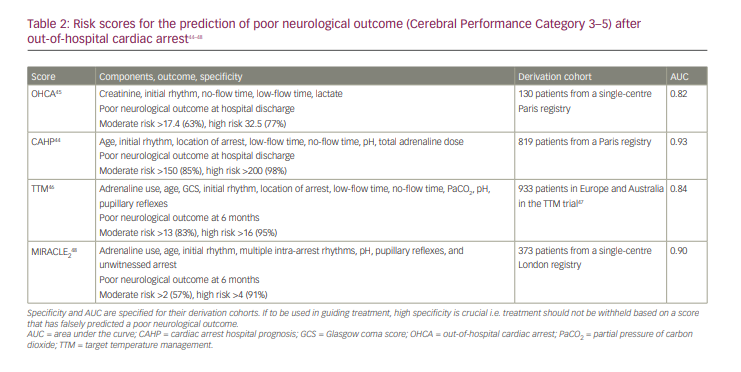
The most recent of these is MIRACLE2, which requires prospective evaluation; however, it performed as well as or better than other scores in predicting poor neurological outcome within its validation cohort. Its key advantages are application on arrival to a heart attack centre, practicality of use with only seven variables (a score range of only 0–10) and avoiding unreliable measures of low-flow and no-flow times.48 Possible limitations include the generalizability of the score to an all-comers population, with both derivation and validation carried out in large tertiary centres, over half presenting with STEMI and 70% with an initial shockable rhythm. As with all risk scores, we must be careful not to create a self-fulfilling prophecy where withholding care makes a poor outcome inevitable. The role of these risk scores in clinical practice, as well as what would be considered an ‘acceptable’ risk of poor neurological outcome with which to guide treatment decisions, have yet to be defined.
Coronary culprits or coronary occlusions?
If there is to be a benefit from immediate coronary angiography, it must lie in those with occlusion and/or ongoing ischaemia, rather than merely a ‘culprit’ lesion. How can we improve our chances of identifying these patients? COACT and TOMAHAWK have shown a stark difference in the rates of culprit lesions and vessel occlusion at angiography compared with previous observational data. The presence of an initial shockable rhythm has been shown to be a predictor of both survival and a coronary cause for cardiac arrest.49 However, if COACT is believed to be truly representative of an all-comers population, then VF/VT alone appears to be insufficient for predicting a culprit lesion or vessel occlusion.
The ACS2 score uses the presence of angina, congestive heart failure, a shockable arrest rhythm or the presence of ST elevation in two contiguous leads (2 points) to predict the presence of a culprit lesion.50,51 Unfortunately, the absence of ST elevation reduces the area under the curve for this score to 0.74. This study did not exclusively involve comatose patients, but we can infer that a collateral history revealing the presence of angina or heart failure symptoms prior to cardiac arrest increases the likelihood of a coronary culprit lesion.
At the simplest level, we have the post-resuscitation ECG. It is important to consider that exclusion of ST elevation leaves a wide spectrum of possible ECG findings, from dynamic and widespread ST changes to the completely normal ECG. In addition, the post-resuscitation ECG may be challenging to interpret, as it is often subject to metabolic derangement and ischaemia due to global low flow (as opposed to a coronary culprit) in the immediate post-ROSC phase. A retrospective study of 370 patients suggested that the false-positive rate for ECGs that meet STEMI criteria was significantly higher within the first 7 minutes after ROSC versus later ECGs (although the endpoint for this study was the subjective need for PCI rather than thrombolysis in myocardial infarction [TIMI] flow).52 Conversely, it has been suggested that typical patterns of cardiac ischaemia on the initial ECG (classical STEMI; ST elevation in aVR with diffuse ST depression elsewhere; modified Sgarbossa criteria; posterior infarction) maintain their predictive accuracy for thrombotic occlusion, even in the presence of metabolic abnormalities.53
It is worth noting the prospectively defined observational NORCAST (Norwegian cardio-respiratory arrest study; ClinicalTrials.gov identifier: NCT01239420) study, which performed coronary angiography on 257 sequential resuscitated OHCA patients at a single centre, irrespective of ECG findings, provided there was no clear non-cardiac cause.43 In this study, an acute lesion was defined as the angiographic presence of thrombus or the ability to easily wire through the lesion. In 210 patients, acute TIMI 0–1 flow was identified in 11% of patients even in the absence of any significant ST changes or bundle branch block on their first ECG after ROSC.43 This observational study remains significant even in the wake of COACT and TOMAHAWK, providing an insight into the true prevalence of acute coronary occlusion after OHCA and demonstrating that the initial 12-lead ECG alone appears insufficient for predicting coronary anatomy.
Evaluation and management of the out-of-hospital arrest patient
In the absence of definitive evidence and faced with the diagnostic and prognostic uncertainty of the post-cardiac arrest patient, clinicians making decisions based on anecdotal experience can lead to either extremes of unfounded nihilism or speculative optimism and potential for waste of limited resources. Immediate activation of the catheter laboratory affects the provision of services for other patients, and out-of-hours work induces team fatigue and a potential loss of workforce for the following day. These effects will be most marked in small and low-volume centres. While observational studies have previously suggested a benefit for early angiography, we now have randomized evidence to guide decision making. Although there are limitations to both COACT and TOMAHAWK, the message from both is consistent: in the absence of ST elevation or cardiogenic shock, a routine strategy of immediate coronary angiography has no benefit.
Admission to a cardiac arrest centre with access to all diagnostic and therapeutic services on site, as well as high-volume provider experience in the management of OHCA, is recommended,54 and allows immediate multidisciplinary evaluation of these patients (Figure 4).33 ST elevation, cardiogenic shock and/or suspected myocardial infarction in the awake patient should proceed directly to immediate catheter laboratory activation. This should also be the case in the comatose patient with ST elevation or cardiogenic shock unless they are clearly moribund with multiple adverse features. Given the difficulties in early neuro-prognostication, the threshold for not proceeding to the catheter laboratory in these cases should be set high.
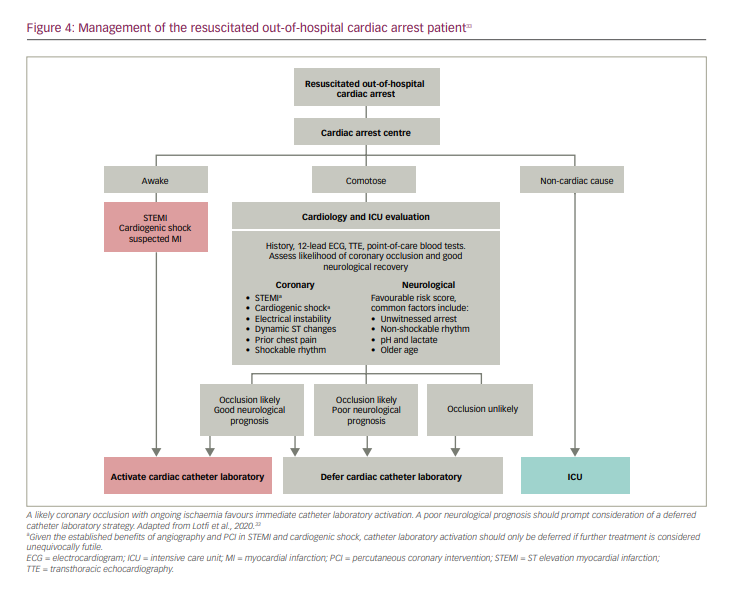
In the comatose patient, an assessment of the likely neurological outcome, as well as the likelihood of ongoing coronary occlusion, should be made. For the vast majority of patients, an initial admission to the ICU for stabilization and diagnostic evaluation (including serial ECGs) is appropriate. Subsequent development of ST elevation, widespread ST changes, haemodynamic or electrical instability would then favour immediate (<2 hours) catheter laboratory activation, which is largely in line with current guidance.5 In the absence of these features, a delayed angiography strategy until neurological prognostication can be carried out and alternative diagnoses excluded seems reasonable (Figure 4).33
Future directions
The DISCO study (Direct or subacute coronary angiography inout-of-hospital cardiac arrest; ClinicalTrials.gov identifier: NCT02309151) is recruiting over 1,000 comatose patients in Sweden following resuscitated cardiac arrest without ST elevation or cardiogenic shock, and randomizing them to immediate versus delayed (12–24 hours) angiography.55 This has expanded in 2021 to all tertiary centres in Denmark who will also recruit non-comatose patients into the separate, similarly sized DISCO-no-COMA study (Direct or subacute coronary angiography in patients with out of hospital cardiac arrest without coma;ClinicalTrials.gov identifier: NCT04876222).56 All patients after a resuscitated cardiac arrest who do not proceed to immediate coronary angiography will therefore be eligible for enrolment into a trial, providing the largest and most comprehensive study in this population to date (Table 3).34,36,42,55–59
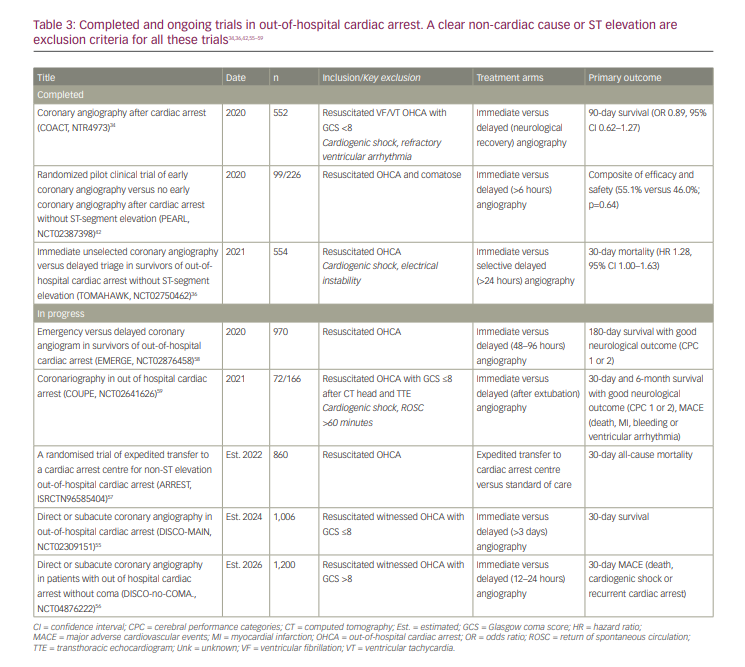
The ARREST trial (A randomised trial of expedited transfer to a cardiac arrest centre for non-ST elevation out-of-hospital cardiac arrest; ClinicalTrials.gov identifier: NCT03872960) will test whether changes at a network level can potentially form an earlier link in the chain of survival, by testing expedited transfer to the cardiac catheter laboratory of a heart attack centre versus standard care.57 Although there remains uncertainty about which components of post-cardiac arrest care improve outcomes, the authors hypothesize that treatment within a tertiary centre with provider experience and the full range of diagnostic and interventional services available will reduce overall mortality.
The next 5 years promise a wealth of data and further answers to help deliver the optimal treatment, and to definitively establish the role of the catheter laboratory after resuscitated OHCA.