Atherosclerosis is a leading health problem worldwide, especially with an aging population and the increasing incidence of prediabetic and diabetic conditions. In addition to coronary artery disease (CAD) and stroke, peripheral arterial disease (PAD) is a major cause of disability. The use of endovascular treatment for symptomatic lower extremity atherosclerotic disease has increased and spread worldwide, and is considered as an important therapeutic component in addition to medical therapies. Although PAD treatments have advanced over the past decade, endovascular treatments for the lower extremity arteries are still limited by relatively high restenosis rates and lack of sustained benefit.1,2 Drug-coated balloons (DCBs) have emerged as a novel approach for patients with PAD and also for patients with in-stent restenosis (ISR) after stent-based treatment of CAD, and to a lesser extent, for coronary de novo lesions. DCBs treat the atherosclerotic lesion with anti-proliferative drugs similar to drug-eluting stents (DES) while ‘leaving nothing behind’, i.e., no permanent metallic stent scaffold. This concept is attractive not only because of the freedom from late complications related to persistent foreign-body reaction (stent thrombosis and ISR), but also because it allows for less-invasive treatment of lesions where stent implantation might not be suitable, e.g., at flexion point in peripheral arteries, bifurcation lesions, ostia, small vessels, and ISR lesions.3–6
In this review, we will focus on novel DCB technologies from the standpoint of recent preclinical animal studies and clinical trials. We will review strengths and weaknesses of current and future technologies in this area as well as discuss current clinical controversies regarding the increase in death rates associated with the use of paclitaxel-eluting DCB versus control devices seen in a recent meta-analysis.
Drug-coated balloons – mechanisms of drug transfer and effectiveness
The main causes of restenosis after balloon angioplasty are acute vessel recoil and late smooth muscle-cell proliferation. Although stent implantation prevents acute recoil, it also causes excessive proliferation of smooth muscle cells and proteoglycan deposition. Both require anti-proliferative drugs for inhibition of restenosis. In general, neointimal growth occurs within the first 7 days to 3 months in humans following the procedure7,8 and therefore, requires drug presence for this period or even slightly longer. Ideally, drug levels should decline thereafter to allow for complete vessel healing. In order to demonstrate efficient performance in the absence of metallic scaffold and polymer, DCBs must deliver and distribute large quantities of anti-proliferative drug to the vessel wall for an extended period of time and maintain therapeutic drug levels in the vessel wall. Transporter excipients and drug selection play an essential role in the formulation of effective DCB.
Excipients
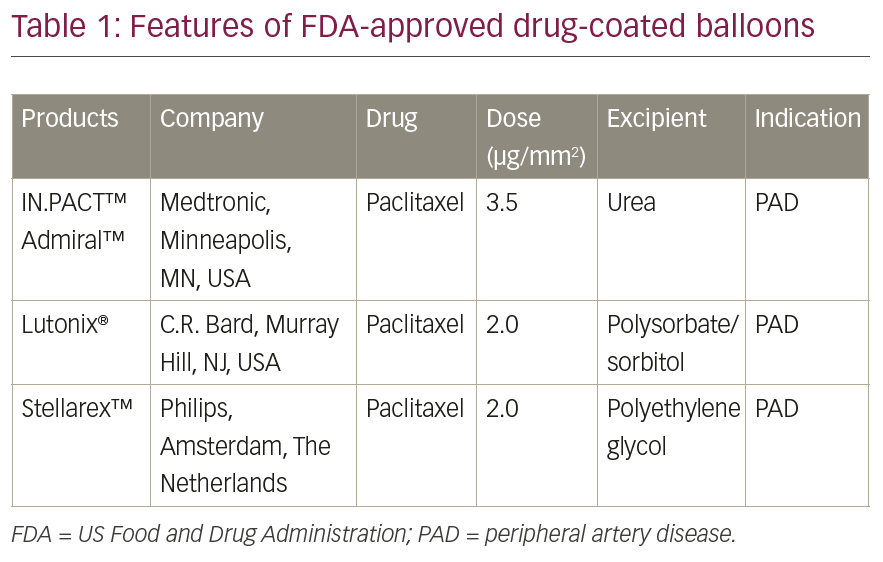
One of the most important innovations in DCB technology was the adoption of transporter excipients to facilitate drug delivery to the vessel wall. Each DCB has unique excipients (e.g., urea, polysorbate/sorbitol, polyethylene glycol), which have distinct qualities (Table 1). To date, all DCBs for PAD approved by the US Food and Drug Administration (FDA) contain paclitaxel (Table 1), the rationale for which we will discuss in detail below. Without excipients, paclitaxel migration from the DCB into the tissues is limited. In one preclinical study, comparing a paclitaxel plus excipient (i.e., iopromide) DCB to a paclitaxel-coated DCB (without excipient) the rate of drug–tissue concentration was significantly higher for the paclitaxel-coated DCB plus excipient.9 Hydrophilic excipients allow higher tissue concentrations and thus more effective restenosis prevention. In general, paclitaxel is a lipophilic poorly water-soluble compound. Excipients can help solubilisation of the compound and the transportation of paclitaxel into tissues.10 Another preclinical study showed that approximately 25–35% of the paclitaxel-loaded on the balloon with excipient coating was lost into the bloodstream during the procedure.11 At the same time, hydrophilic excipient coatings facilitate higher drug–tissue levels, they also allow high wash-off of the coating before their delivery to the target site. As discussed later, the possibility of harmful effects to downstream non-target organs of paclitaxel debris remains uncertain but such emboli have been detected in preclinical animal models.12,13
Drugs
Currently, paclitaxel is the most widely used anti-proliferative drug for DCBs mainly because of its high-lipophilic characteristics that allow a passive absorption through the cell membranes and persistent effect inside the target vessel wall.12 Although both the amorphous and crystalline forms of paclitaxel have been used on DCBs, the balance of crystalline to amorphous forms affects the pharmacokinetic (PK) behaviour of the drug and thus impact neointimal formation and healing. In one animal study, DCBs were loaded with either the crystalline or amorphous forms of the drug in equal doses (3 μg/mm2), deployed in pigs, and arterial wall PK levels of the drug examined. Although both formulations achieved similar arterial paclitaxel levels 1 hour after deployment, the crystalline forms retained more drug at both 24 hours and 28 days.14 For commercially available DCBs in the US, dosages of paclitaxel range from 2.0–3.5 μg/mm2 (Table 1).
In the DES market for femoropopliteal artery disease, paclitaxel is also primarily used in the two Conformité Européene (CE)-marked and FDA-approved self-expanding DESs. These DES’s have shown excellent clinical outcomes for up to 5 years (Zilver® PTX®; Cook Corporation, Bloomington, Indiana, USA) and 3 years (Eluvia™; Boston Scientific, Marlborough, Massachusetts, USA).15,16 On the other hand, DESs employing sirolimus or its analogues (e.g., everolimus) failed to show superiority against bare metal stent for the treatment of femoropopliteal artery disease.17,18 Discrepancies in the results between paclitaxel and sirolimus and its analogues used on DESs may be due to more than just differences in the pharmacologic agents themselves. Self-expanding stents continuously expand and cause inter-strut distance increase over time, resulting in decreased drug availability in areas between the struts.19 Sirolimus and its analogues, when loaded onto balloon-expandable DESs, showed mostly successful outcomes for below-the-knee lesions with focal stenosis or occlusion at 1-year follow-up.20,21 However, most of these lesions were short and thus not necessarily representative of clinical practice. When treating below-the-knee lesions, stenting should generally be avoided in arterial segments which might limit future therapeutic options such as landing zones for potential bypasses. Evidence on whether this technology can be effective for longer lesions, where more than one DES would be required, is also still lacking.
Sirolimus and its analogues offer potential benefit over paclitaxel in terms of safety and efficiency; sirolimus shows wider safety therapeutic range, and a greater anti-restenotic and anti-inflammatory effect than paclitaxel, which is one major reason why paclitaxel-eluting stents are no longer available for use in coronary arteries.22,23 Although paclitaxel has advantages of better tissue absorption and retention as compared to sirolimus, improvement of excipients, formulation (i.e., crystalline sirolimus), and encapsulation using polymer-based micro- and nanoparticle technologies may allow sirolimus to have a potential for DCB use. Recently, several clinical and preclinical studies have shown the efficiency and safety of sirolimus DCBs.24–26 Furthermore, as we will discuss below, recent safety concerns about paclitaxel DCBs have brought to the forefront the need for sirolimus DCBs.27
Finally, there are fewer data on drug-eluting/coated devices used for PAD rather than CAD. One should be careful when extrapolating data from one area to the other considering the differences between the pathophysiology of PAD and CAD.
Pathology of drug-coated balloons in preclinical models
Despite the effectiveness of excipient, the drug-coating integrity of DCBs is variable. In one preclinical animal study, paclitaxel loss on the way through the guiding catheter and blood was in the range of 30%.11 Loss of paclitaxel into the body, not necessarily at the target site, allows for potential of downstream embolisation of the drug/excipient. Whether such events could be responsible for unfavourable outcomes remains uncertain but is an undesired consequence of current paclitaxel DCB technologies. Several case reports have been published suggesting adverse effects, mainly in the forms of vasculitis/panniculitis, after DCB treatment for PAD and CAD.28–31
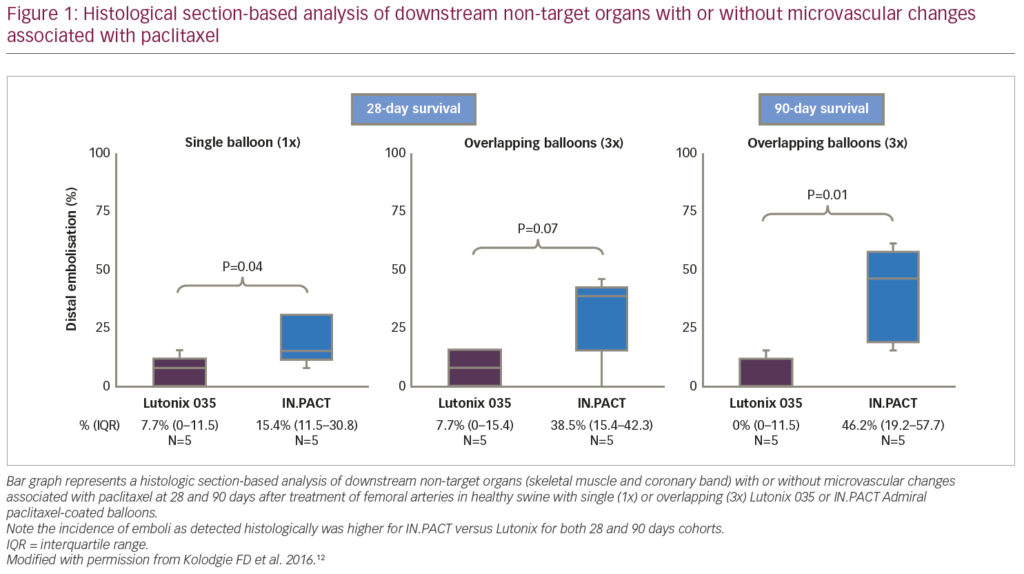
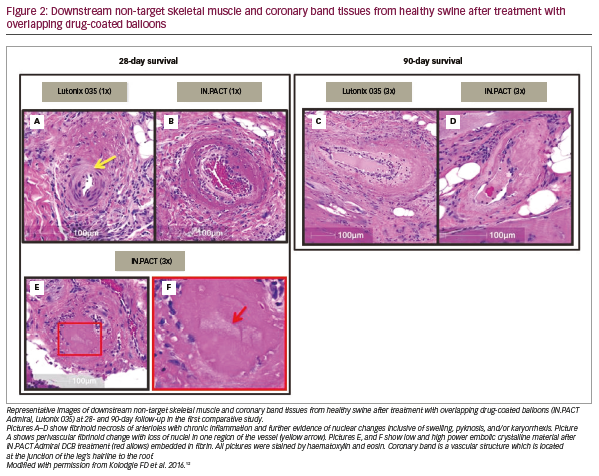
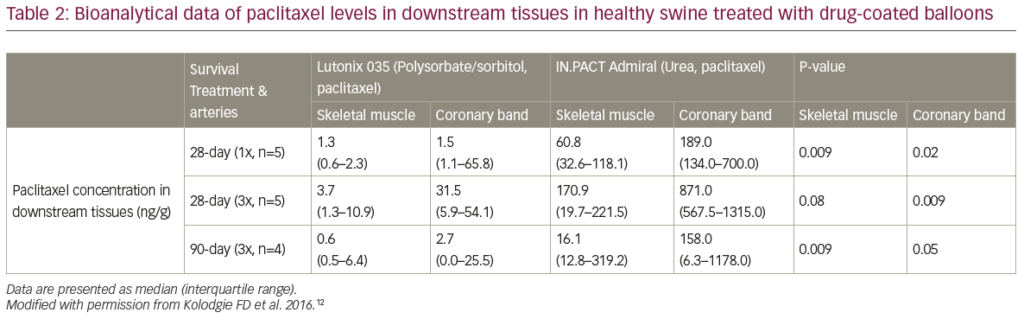
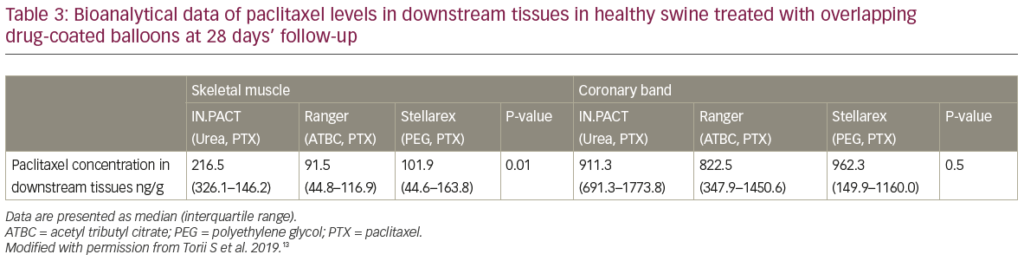
We have previously conducted two consecutive preclinical studies to evaluate the downstream effect after DCB treatment in healthy swine iliofemoral arteries.12,13 In the first study, histological changes and drug concentration in downstream skeletal muscles and the coronary band (a vascular structure which is located at the junction of the leg’s hairline to the hoof) were evaluated at 28 and 90 days following the procedure with single or overlapping (3x) IN.PACT™ Admiral™ DCB (paclitaxel 3.5 μg/mm2) (Medtronic, Minneapolis, MN, USA) versus Lutonix® 035 DCB (paclitaxel 2.0 μg/mm2) (C.R. Bard, Murray Hill, NJ, USA) in healthy swine iliofemoral arteries.12 The percentage of sections with paclitaxel-associated effects were significantly higher for IN.PACT than for Lutonix (Figure 1). Embolisation of crystalline-like material was only observed in the IN.PACT arm involving five out of 195 sections, but fibrinoid necrosis of skeletal muscle arterioles with chronic inflammation was seen for both devices, suggesting paclitaxel-induced injury (Figure 2). Differences in drug concentration in downstream tissues between the two devices were many-fold higher for the IN.PACT than the Lutonix (Table 2). In a separate study, we also compared the downstream effect among three different DCBs (IN.PACT Admiral, Ranger™ [paclitaxel 2.0 μg/mm2] [Boston Scientific, Marlborough, MA, USA], and Stellarex™ [paclitaxel 2.0 μg/mm2] [Philips, Amsterdam, Netherlands]) in 12 healthy swine using similar methods as used in the previous study.13 Histological analysis showed that the prevalence of downstream vascular changes was equivalent for all three DCBs (Table 3). Paclitaxel concentration in downstream skeletal muscle beds was significantly higher in the IN.PACT arm as compared with Ranger and Stellarex, but no differences were observed in the coronary band (Table 3). These two preclinical studies suggest that the various features of DCBs (i.e., drug formulation, dose, and excipient) may affect the degree of distal embolisation. Thus, even in the absence of firm clinical data suggesting adverse effects from emboli, further development of DCB technology with less downstream embolic effects, is warranted. It remains uncertain how these findings in healthy juvenile animals translate into aged humans with multiple co-morbidities and compromised arterial supply in lower extremities.
Clinical study outcomes
Coronary artery disease
European guidelines recommend DCB use for coronary artery ISR lesion as a class I treatment.32 Although DES is a very effective mode of revascularisation for CAD, there remain important limitations to this type of technology such as stent thrombosis, neo-atherosclerosis, and late restenosis. DCBs may have the potential to overcome some of these. Primary advantages include absence of requirement for long-term dual antiplatelet therapy and the treatment of lesions where DES are unsuitable, e.g., bifurcation and small vessel.4–6
The treatment of ISR lesions is still considered a challenging problem despite the availability of DESs.33 Elgendy et al. conducted a meta-analysis comparing the target-lesion revascularisation (TLR) with a DCB or a second-generation DES (everolimus-eluting stent) for the treatment of ISR lesion.6,34 A total of 1,363 patients from seven randomised controlled trials were included. All randomised controlled trials compared DES with DCB for any ISR (i.e., bare metal stent or DES) lesions. Although the short-term primary endpoint (i.e., TLR) in the DCB group was higher than in the DES group (11.4% versus 5.6%; relative risk [RR], 1.83; 95% confidence interval [CI], 1.07–3.13, p=0.03) at a mean of 27 months, the DCB group showed no difference from the DES group regarding major adverse cardiac event (MACE; TLR, myocardial infarction, stent thrombosis, and all-cause death). These data support the use of DCBs for the treatment of ISR.
DCBs may also be used for the treatment of small vessel CAD which might not be suited for a DES due to high restenosis rates. The BASKET-SMALL 2 study evaluated the effectiveness and safety of DCB use in small vessel de novo coronary lesions. The study was a multicentre, randomised controlled trial including 758 patients with de novo small vessel CAD (diameter <3 mm). Patients were randomly assigned to the paclitaxel DCB versus second-generation DES. Lesion preparation was compulsory before randomisation. If angiographic findings did not meet the inclusion criteria, subjects were excluded from the trial.
The primary endpoint was determined by non-inferiority in terms of MACE (cardiac death, non-fatal myocardial infarction and TLR) after 12 months. The incidence of MACE after 12 months revealed no significant difference between both groups (DCB versus DES; 7.3% versus 7.5%, p=0.918), with no difference seen for each component of the MACE. In a similar fashion as for ISR lesions, DCB usage for small vessel lesions has shown advantages over stenting as long as optimal lesion preparation is achieved successfully.5 Recently, Magic Touch® sirolimus-coated DCB (Concept Medical Inc, Tampa, Florida, USA) was granted “breakthrough device designation” from the FDA for the treatment of coronary ISR in April 2019. This may become the first DCB approved for CAD treatment in the USA. However, there is little evidence that sirolimus DCB is safer and more effective than paclitaxel DCB, especially long-term follow-up. One should keep in mind that sirolimus DCB needs to be independently tested in randomised clinical trials before its safety and effectiveness can be said to be better than paclitaxel DCBs.
Peripheral arterial disease
At present, DCBs are widely used for the treatment of symptomatic PAD not only in European countries but also in the USA. According to the latest European Society of Cardiology (ESC) guidelines, the use of drug-coated devices is recommended for ISR or short segment stenosis (<25 cm) in femoropopliteal lesions, as class IIb treatment.35 Femoropopliteal arteries are the most dynamic and the longest arteries in the human body and cover hip and knee flexion points. Therefore, these vessels receive multiple external stresses such as torsion, compression, flexion, and extension,36 and such stresses may cause stent fractures.37 Although recent DES technology has improved, the prevalence of both stent fracture (1.9% in the 5-year follow-up) and 5-year TLR rate (17%) remains high,15 and inferior to that reported for coronary arteries (approximately 10%).38 Thus, DCBs are an attractive choice for treatment because they may be able to overcome some of these limitations while allowing for future treatment option such as surgical bypass procedures.
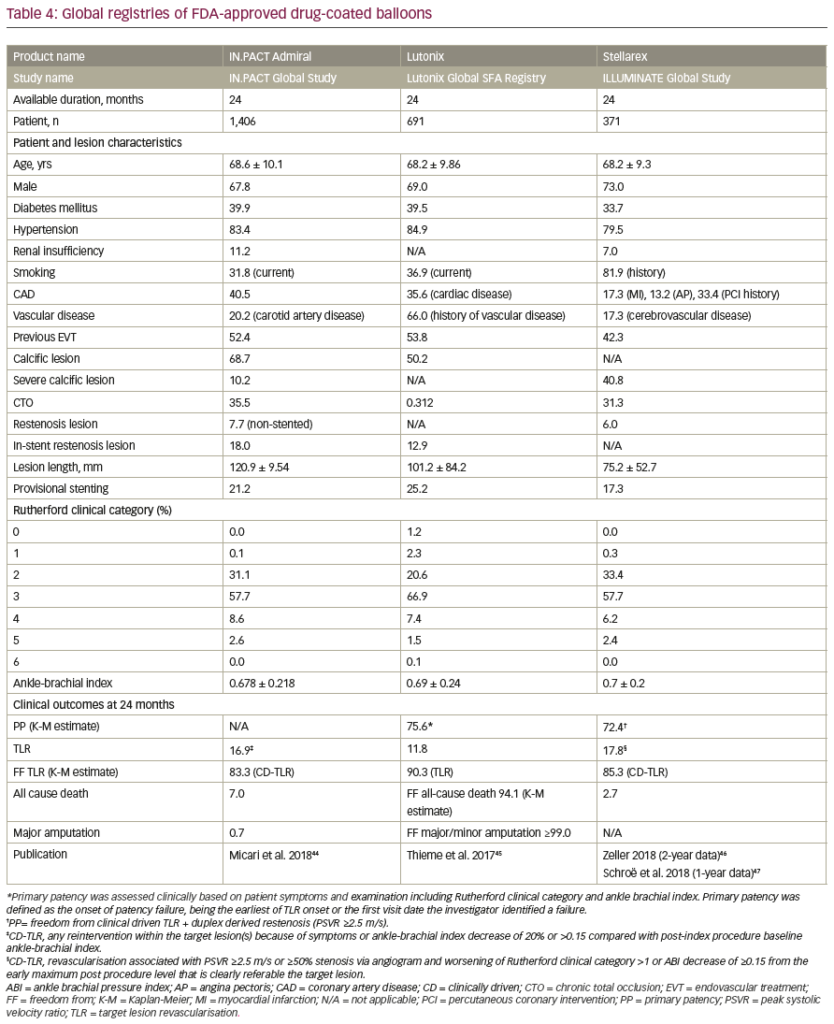
There are three FDA approved DCBs for PAD treatment in the USA (Table 1). Each of these DCBs demonstrated in randomised controlled trials safety and efficacy compared to standard percutaneous transluminal angioplasty.39–43 To test the validity of the result of randomised controlled trials at a worldwide level, each manufacturer also conducted large global registries which include patients with multiple comorbidities and more complex lesions (e.g., patients with critical limb ischaemia, chronic total occlusion and long segment lesion) than what was allowed in randomised controlled trials (Table 444–47). All three registries included a larger number of patients with comorbidities and complicated lesions, and have shown favourable results.
Current drug-coated balloon controversy
A recent meta-analysis of randomised controlled trials, published in the Journal of the American Heart Association, demonstrated increased mortality rate at 2 and 4–5 years in patients with PAD treated with paclitaxel-coated balloons and paclitaxel-eluting stents compared to patients treated with control devices (plain old balloon angioplasty or bare metal stent).27 In this meta-analysis, the authors investigated the results of randomised controlled trials using paclitaxel DCBs and DESs in femoropopliteal arteries. Twenty-eight randomised controlled trials with 4,663 patients were analysed. All-cause patient death at 1 year (28 randomised controlled trials with 4,432 cases) was comparable between paclitaxel-coated devices and control arms (2.3% versus 2.3% crude risk of death; RR 1.08; 95% CI, 0.72–1.61). However, all-cause death at 2 years (12 randomised controlled trials with 2,316 cases) and 4–5 years (three randomised controlled trials with 863 cases) was significantly increased in the case of paclitaxel versus control cases (7.2% versus 3.8% crude risk of death; RR, 1.68; 95% CI, 1.15–2.47, and 14.7% versus 8.1% crude risk of death; RR, 1.93; 95% CI, 1.27–2.93, respectively). As a result of meta-analysis, unfavourable criticism of paclitaxel attracted much attention and gave rise to a discussion about the safety of such devices. Subsequently, the FDA issued a statement to healthcare providers in both January and March 2019.48 The FDA conducted its own preliminary analysis of long-term follow-up data of the pivotal premarket randomised trials for paclitaxel-coated devices indicated for PAD and identified a potentially concerning signal of increased long-term mortality in patients treated with paclitaxel-coated devices versus patients treated with uncoated devices. Among the 975 patients in three trials, there was an approximately 50% increased risk of mortality in patients treated with paclitaxel-coated devices versus those treated with control devices (20.1% versus 13.4% crude risk of death at 5 years). Although these data did not show the relationship between paclitaxel exposure and specific causes of death, the FDA is continuing to evaluate the increased long-term mortality signal as well as the overall benefit-risk profile of these devices. The current FDA recommendations are to conduct ‘…diligent monitoring of patients who have been treated with paclitaxel devices and to discuss risks and benefits of all available PAD treatment options with the patients. For most patients, alternative treatment options to paclitaxel devices should generally be used until additional analysis of the safety signal has been performed’.
Despite these rather clear recommendations to avoid paclitaxel coated/eluting devices, there are limitations to the Katsanos’s meta-analysis that deserve special attention.27 Most of the randomised controlled trials did not report the actual cause of death which is necessary to determine potential causal links with paclitaxel device use and mortality since the side effects of paclitaxel are well known from the oncology literature. Although a significant difference in mortality rate between the paclitaxel arm and the control arm was shown from 2 years and 4–5 years post-procedure, the number of patients included in the study dropped sharply from the 1-year time point (28 studies) to both 2- (12 studies) and 5-year (two studies) timepoints. It remains uncertain if more patients were included (as they become available) in the later timepoints whether this would affect the overall results. For instance, for the 5-year result of Zilver PTX trial, published in Circulation 2016, 28% of patients were lost to follow-up or withdrew.49 Moreover, the meta-analysis used the intention-to-treat principle without accounting for the rate of crossover to paclitaxel coated/eluting devices. Thus, the relationship between exposure and outcome could be confounded by significant crossover in the percutaneous transluminal angioplasty arm of every trial. Furthermore, the study only demonstrated an association between paclitaxel exposure and death, but such an analysis cannot prove cause and effect, which is a crucial limitation of the study.
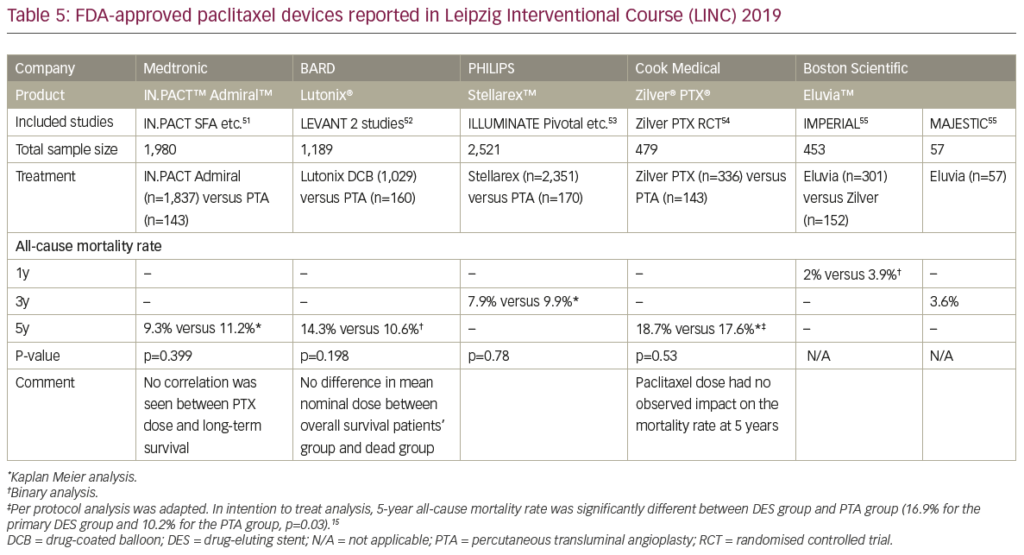
Schneider et al. published independent patient-level meta-analysis of paclitaxel DCB treatment for patients with symptomatic PAD.50 In this analysis, the authors examined 1,980 patients enrolled from four trials of IN.PACT Admiral DCB. After clearly categorising the cause of death, the analysis was conducted on patient data up to 5 years. The amount of paclitaxel dosage to patients was documented and patients were divided into three groups (lower, mid, and upper tertile) based upon paclitaxel exposure; mortality rates were calculated according to the dose of paclitaxel. There was no statistically significant difference in all-cause mortality among three groups up to 5 years. Other device manufacturers also reported patient-level analysis of their product’s trials at Leipzig Interventional Course (LINC) 2019. All devices did not show any relation between paclitaxel exposure and mortality (Table 5).51–5 Secemsky et al. have reported a large Medicare- and Medicaid-based analysis of the relationship between paclitaxel device use and mortality.56
They compared 5,989 patients with PAD who received treatment with paclitaxel devices to 10,571 patient with PAD who received treatment with standard percutaneous transluminal angioplasty. Median follow-up was 389 days (interquartile range, 277–508 days), with 8,450 patients (51%) presenting with critical limb ischaemia. Multivariate analysis did not show the relationship between paclitaxel device use and mortality (adjusted hazard ratio, 0.97; 95% CI, 0.91–1.04; p=0.43). Although shorter follow-up duration and patients with higher critical limb ischaemia might conceal the association between paclitaxel device use and mortality, this study also did not confirm the results reported in the Katsanos’s meta-analysis.
Lastly, it must also be acknowledged that paclitaxel is a well-known chemotherapeutic agent used for the treatment of various cancers including breast, ovarian and non-small cell lung cancers for decades. Its toxicities are well understood in the oncology field and include bone marrow suppression, neurotoxicity, alopecia and nausea. Multiple regimens of high doses of paclitaxel are usually required to cause such effects which are mostly seen in dose ranges at or above 175 mg/mm2.57 Doses for DCBs, even when using multiple balloons, do not even come close to this level (i.e., total dose for IN.PACT Admiral 5.0 x 120 mm balloon is 3.8 mg of paclitaxel). Moreover, plasma PK levels for paclitaxel do not ever approach levels high enough to cause bone marrow suppression. Data from the LEVANT I trial (pivotal trial for Lutonix DCB) showed plasma drug levels peaked at 58 ng/ml within minutes and then declined rapidly thereafter became undetectable after 30 hours.58 Thus, another weakness of the Katsanos’s meta-analysis is that no clear explanation for how paclitaxel at doses delivered on DCBs could cause death.
Conclusion
DCBs may provide anti-restenotic benefits for the treatment of atherosclerotic disease without the drawbacks of DES. Nevertheless, there are serious controversies surrounding their use. Although case reports and preclinical studies have shown potential safety concerns of current available paclitaxel DCBs, mainly in the form of emboli to non-target tissues, increased mortality was not seen in preclinical studies required for approval of DCBs. Katsanos’s meta-analyses suggested the association between mortality and paclitaxel device use; however, there does not appear to be a clear mechanism of how DCBs would result in increased mortality of treated patients. Further patient-level analyses are needed to determine whether the current concern over DCB safety is indeed warranted. Nonetheless, the development of safer and more effective DCBs should continue as they represent a unique technology which has distinct benefits for patients.