Background: DC cardioversion (DCCV) is commonly used to assess the symptomatic benefit of sinus rhythm (SR) when deciding whether to offer further rhythm control therapies such as AF ablation. However, acute failure of DCCV to achieve SR and early reversion to AF are common. This makes assessment of the symptomatic benefit of SR very difficult and many patients undergo repeat DCCV, often while taking amiodarone. We sought to identify factors associated with acute DCCV failure and early reversion to AF, and whether amiodarone therapy before first DCCV could reduce the failure rate in high risk populations
Methods: Retrospective analysis of electronic medical records of patients undergoing DCCV during a 12-month period from Jan-Dec 2017
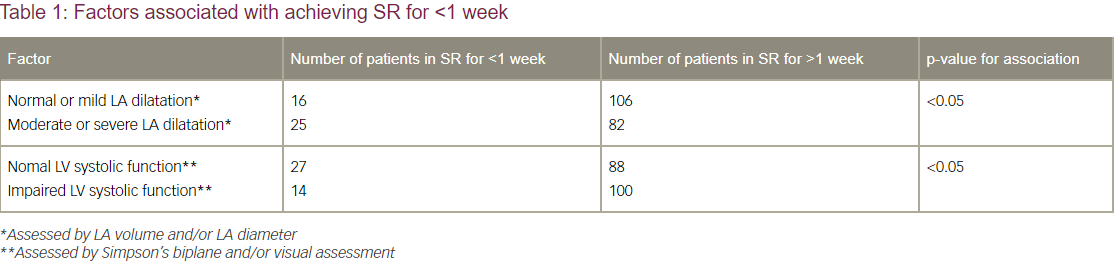
Results: 239 patients underwent DCCV. Mean age 68 (range 31-89), 68% male. 68% underwent first DCCV, 23% DCCV 2, 7% DCCV 3, and 2% DCCV 4. Follow-up and echocardiographic data was available for 229/239 (96%) patients. 42/229 (18%) achieved SR for <1 week (25/42 acute DCCV failure, 17/42 reversion to AF after <1 week). Factors assessed for association with achieving SR for <1 week were: age >65, documented hypertension, documented obesity, AF duration >12 months, LA dilatation, LV systolic impairment and mitral regurgitation. Only two of these factors were significantly associated with achieving SR for <1 week: the presence of moderate / severe LA dilatation compared to normal sized / mildly dilated LA, and unexpectedly the presence of normal LV systolic function compared to impaired LV systolic function (see Table 1). There was no significant difference in the number of patients taking amiodarone within these two groups. 19 patients had previously undergone DCCV 1 and achieved SR for <1 week, who then underwent DCCV 2 while taking amiodarone. 12/19 (63%) of these patients then achieved SR for >1 week. There was no difference in LA dilatation or LV systolic function in this group compared to the general DCCV 1 population.
Conclusion: Achieving <1 week of SR after DCCV was common. It was associated with the presence of LA dilatation and, unexpectedly, normal LV systolic function. The LV function result may have been confounded by difficulty in echocardiographic assessment of LV systolic function in the presence of AF, but does add support to offering rhythm control therapies to selected patients with LV systolic impairment. Among patients who had previously achieved <1 week of SR after DCCV 1 who underwent DCCV 2 on amiodarone, most achieved SR for >1 week. This suggests that temporary amiodarone therapy before DCCV 1 in patients at high risk of early reversion to AF, such as those with LA dilatation, could increase the number who achieve >1 week of SR. This would reduce the requirement for second DCCV and may reduce time to AF ablation in this patient group.