Coeliac disease (CD) is a systemic disease of inappropriate immune response to gluten, which affects up to 1% of the population in the Western world.1 Gluten proteins are found in dietary products containing wheat, rye, barley and related grains. In patients who carry the human leukocyte antigen (HLA)-DQ2 and HLA-DQ8 alleles of the major histocompatibility complex class II, peptides derived from the incomplete digestion of gluten bind to HLA-DQ2 and HLA-DQ8; induce the inflammatory response of T cells in the small intestine; and lead to intraepithelial lymphocytosis, crypt hyperplasia and villous atrophy.2,3 Although CD primarily affects the small intestine and manifests in diarrhoea, abdominal discomfort/pain and weight loss, CD can affect every tissue in the body.4 Systematic CD manifestations include anaemia, arthritis, dermatitis herpetiformis, delayed puberty, infertility, peripheral neuropathy and decreased bone density.5 Complications of untreated CD may include psychiatric and neurologic disorders including ataxia, epilepsy and seizures, schizophrenia and depression.6
Iron-deficiency anaemia is the most common non-gastrointestinal manifestation of CD. Hence, a suspicion of CD can be raised in patients with anaemia of obscure origin, especially resistant to oral iron supplementation.7 Concomitantly, CD is associated with a 10% increased risk of cardiovascular disease (CVD) due to the higher prevalence of type 1 diabetes mellitus (T1DM), hypercoagulability, hyperlipidaemia and faster progression of atherosclerosis in patients with CD.8,9 Here we present the case of a young patient with persistent myocardial ischaemia due to anaemia, who was eventually diagnosed with CD.
Case report
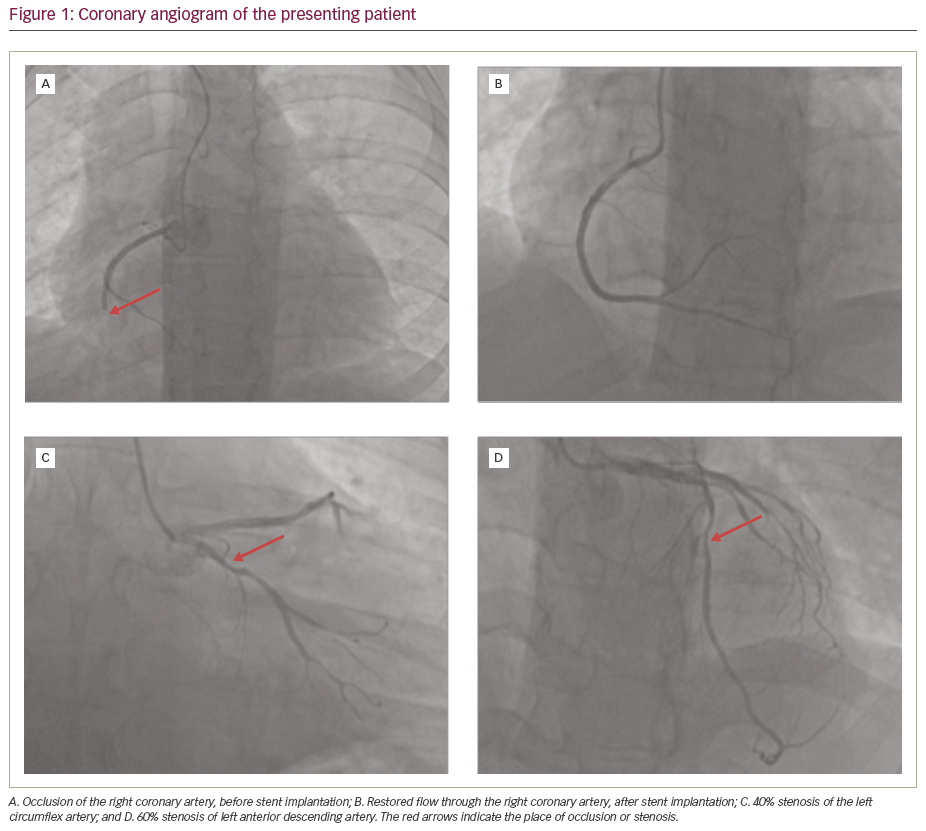
A 36-year-old male smoker was admitted to the cardiology department due to ST-elevation myocardial infarction of the inferior wall. He also suffered from T1DM, complicated by peripheral neuropathy and diabetic foot, hypertension, iron-deficiency anaemia treated with oral iron supplementation, dyslipidaemia, and gastrointestinal reflux disease (GERD). On admission, the patient presented with strong chest pain, without dyspnoea. Physical examination showed tachycardia (105 beats per minute [bpm]) and elevated systolic blood pressure (150/70 mmHg), normal respiratory sounds, no signs of pulmonary or peripheral congestion, and no heart murmur. His skin was pale and the examination revealed a blistering skin rash on the right knee and diabetic foot of the right toe. Emergency coronary angiography (Figure 1) showed occlusion of the right coronary artery, 40% stenosis of the left circumflex artery and 60% stenosis of the left anterior descending artery. Primary percutaneous coronary intervention with stent implantation to the right coronary artery was successfully performed.
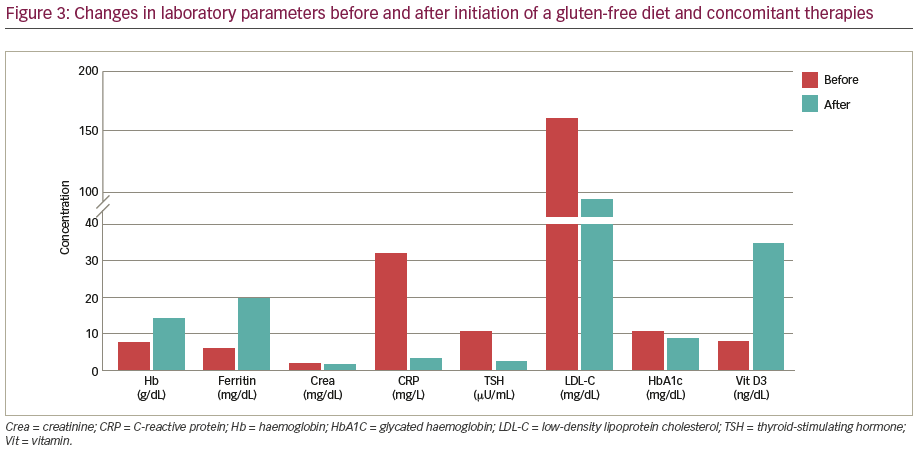
Laboratory tests revealed microcytic anaemia with haemoglobin level of 7.8 g/dL, elevated concentration of cardiac troponins (57 ng/mL), creatinine (1.9 mg/dL, C-reactive protein (CRP; 32 mg/L), low-density lipoprotein cholesterol (LDL-C; 160 mg/dL), triglycerides (230 mg/dL) and glycated haemoglobin (HbA1c; 10.7 g/dL), and primary hypothyroidism (thyroid-stimulating hormone [TSH] 10.6 μU/mL, free thyroxine [fT4] 4.5 pmol/L). Concomitantly, iron (21 μg/dL), ferritin (6 ng/mL) and vitamin D3 (8 ng/mL) deficiency were observed.
Despite the successful intervention, the patient presented with recurrent chest pain, persistent tachycardia up to 120 bpm, and syncope. In the absence of new troponin elevation and new signs of ischaemia on electrocardiogram, stent thrombosis was unlikely. However, coronary angiography was repeated and lesions in the left circumflex artery and left anterior descending artery were proved insignificant using fractional flow reserve evaluation. The control laboratory tests revealed a further decrease in haemoglobin level to 6.5 g/dL after the percutaneous coronary intervention and 2 units of erythrocyte concentrate were immediately transfused, resulting in the haemoglobin level rising to 8.7 g/dL. An emergency gastroscopy with duodenoscopy and coloscopy revealed no tumour and no overt source of bleeding. However, flat duodenal mucosa was observed on duodenoscopy and a suspicion of CD was raised. The duodenal biopsy had to be postponed due to mandatory dual antiplatelet therapy with acetylsalicylic acid and clopidogrel to prevent stent thrombosis and recurrent myocardial infarction. The antibodies against tissue transglutaminase in the IgA class were positive. Meanwhile, other causes of anaemia were investigated. Laboratory tests including cancer antigens (CA 15-3, CA 19-9, CA 125, carcinoembryonic antigen, alpha-fetoprotein, prostate-specific antigen) and markers for autoimmunity (rheumatoid factor, anti-neutrophil cytoplasmic antibodies, antinuclear antibodies) were not elevated.
During hospitalisation, the patient was diagnosed with Hashimoto thyroiditis based on the presence of anti-thyroid peroxidase antibodies, and treatment with levothyroxine was initiated. In addition, (i) gluten-free diet was recommended until the final CD diagnosis could be made, (ii) intravenous iron supplementation was started, (iii) anti-hypertensive, hypolipidaemic and hypoglycaemic therapy were intensified, (iv) vitamin
D3 supplementation was started, and (v) the patient was strongly advised to cease smoking.
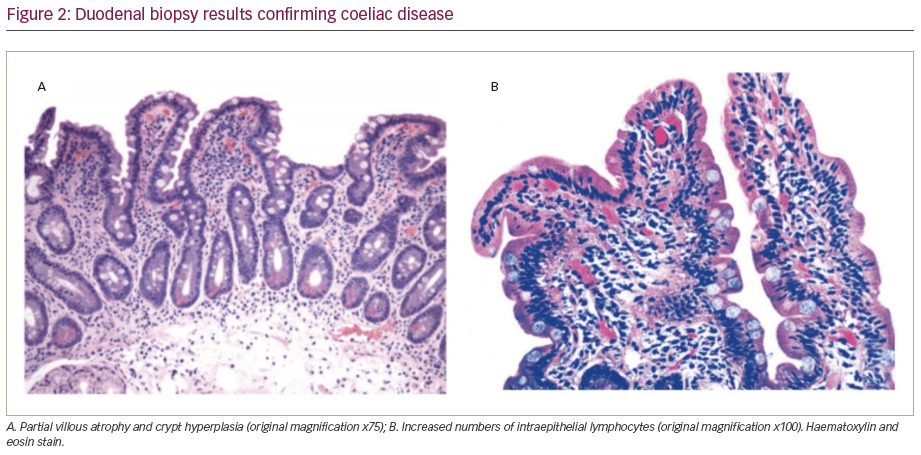
After 1 month, the patient was re-admitted for clinical assessment and duodenal biopsy. During that time, he had a few recurrent episodes of chest pain. At admission, the patient had no complaints. He presented with a heart rate of 98 bpm and blood pressure of 130/85 mmHg, without symptoms of heart failure. Haemoglobin level was 11.4 mg/dL. The duodenal biopsy revealed villous atrophy, crypt hyperplasia and increased numbers of intraepithelial lymphocytes (Figure 2). The diagnosis of CD was confirmed and the strict gluten-free diet, as well as further oral iron supplementation, were recommended.10–12 Two months later the patient presented at the ambulatory clinic with no recurrent chest pain and with normal haemoglobin (14.1 g/dL), ferritin (20 ng/mL) and vitamin D3 (35 ng/dL) levels; stable creatinine (1.8 mg/dL); normal CRP (3 mg/L), TSH (2.5 μU/mL) and fT4 (155 pmol/l); and decreased LDL-C (90 mg/dl), triglycerides (190 mg/dl) and HbA1c (8.9 g/dl; Figure 3). The wound on his right knee (likely dermatitis herpetiformis) was successfully treated. The hypolipaemic and insulin therapy were further optimised.
Discussion
Diagnosis of CD, especially in case of its atypical presentation can be difficult. Our patient had scarce gastrointestinal symptoms, although on detailed interview he admitted to having flatulence and dyspepsia, which had been attributed to GERD. He was diagnosed with iron-deficiency anaemia refractory to oral iron supplementation. The anaemia aggravated the symptoms of myocardial ischaemia, even after a successful coronary revascularisation.
Anaemia has a high prevalence in untreated CD (34%) and may be the only abnormality identified in CD.13,14 Pathophysiology of anaemia in CD is a composite of (i) malabsorption of nutrients in the duodenum due to the reduced absorption area, (ii) chronic inflammation and (iii) occult blood loss due to intestinal wall damage.13,14 Following the introduction of a gluten-free diet, along with intravenous, then oral iron supplementation, the patient had normal haemoglobin levels and iron storage (as indicated by normal ferritin concentration) within only 3 months.15
CD increases the risk of CVD in many ways. Both CD and atherosclerosis are chronic inflammatory processes, mediated by toll-like receptors and interleukin-1 pathways, which are involved in the early steps of arteriosclerotic plaque development.16,17 Thus, low-grade inflammation in the course of CD and other autoimmune diseases can accelerate atherosclerosis.18 Moreover, patients with both T1DM and autoimmune disease have a higher rate of atherosclerosis progression, as compared with patients who have only autoimmune disease.19 Villous atrophy leads to decreased absorption of nutrients, including B vitamins and folic acid. B vitamins and folic acid lower the plasma
level of homocysteine. In the absence of these nutrients, homocysteine levels increase, which is associated with an increased risk of adverse cardiovascular events.18 Villous atrophy may also lead to malabsorption of drugs, including cardioprotective drugs.18 Patients with CD often do not have the traditional cardiovascular risk factors, such as increased body weight, dyslipidaemias or smoking, suggesting that the inflammation is the main trigger of CVD in patients with CD.20 In contrast, our patient presented with many traditional CVD risk factors, including hypertension, dyslipidaemia, poorly controlled T1DM and smoking abuse, making his overall cardiovascular risk extremely high and resulting in an acute myocardial infarction at the age of 36.
The gluten-free diet is proven to alleviate the intestinal inflammation and severity of anaemia in CD, but its overall effect on CV risk is unclear.21 Hence, it was crucial to diminish the traditional risk factors in our patient, including appropriate blood pressure, lipid and glucose control and smoking cessation.
There is an association between CD and other autoimmune diseases, including T1DM, autoimmune thyroiditis, autoimmune hepatitis, Sjogren’s syndrome and psoriasis.22 CD and other endocrine autoimmune diseases share a common genetic background, which are HLA-DQ2 and HLA-DQ8 alleles.23 For example, it is estimated that the prevalence of CD in patients with T1DM is approximately 5%.24 Our patient presented with symptoms of three autoimmune diseases: CD, T1DM and Hashimoto’s thyroiditis. Although specific treatment for each disease is required, a gluten-free diet seems to alleviate not only symptoms of CD, but also symptoms of concomitant autoimmune diseases; although a beneficial effect is not always present.25,26
Finally, about 10% of patients with CD experience dermatitis herpetiformis, which is a blistering and itchy skin rash.24 Our patient had dermatitis herpetiformis on his right knee, which coincided with impaired wound healing in the course of poorly controlled T1DM and the diabetic foot of his right toe. Both dermatitis and diabetic foot wounds alleviated along with the gluten-free diet and glucose control improved. Based on the case of our patient, it is important to critically assess skin abnormalities in patients with autoimmune disease and/or anaemia, as they may help to guide the diagnosis towards CD.
Conclusions
We presented the case of a patient with atypical manifestations of CD, including persistent myocardial ischaemia due to iron-deficiency anaemia, which was refractory to oral iron supplementation. Serological tests and duodenal biopsy allowed the diagnosis of CD to be confirmed, and implementation of a gluten-free diet alleviated both anaemia and the symptoms of myocardial ischaemia.