Case Report
We report on a 53-year-old man with a history of heart failure due to alcoholic dilated cardiomyopathy with severely depressed systolic left ventricular (LV) function. Initial LV ejection fraction was ~20% (Simpson’s biplane method, Loops 1 and 2). At the time of diagnosis his clinical status was New York Heart Association (NYHA) III-IV and he had left bundle branch block. Cardiac resynchronisation therapy (CRT) was initiated after ramipril, bisoprolol and spironolactone had been established for >6 months, intermittent use of loop diuretics was necessary.
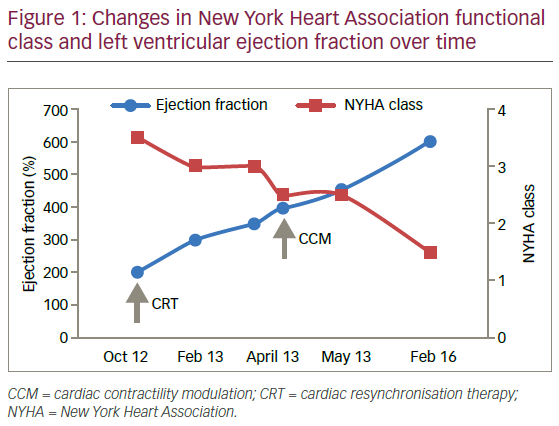
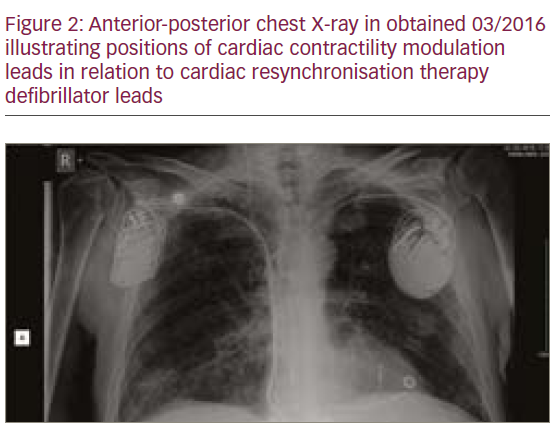
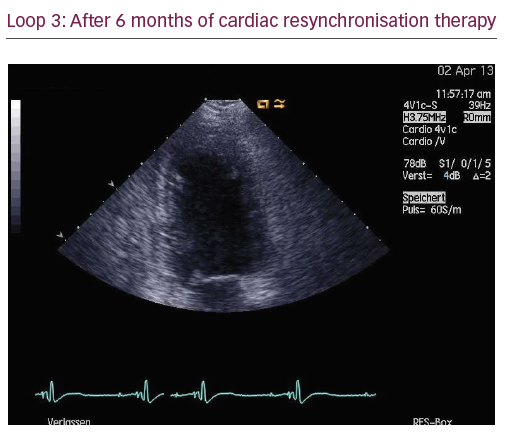
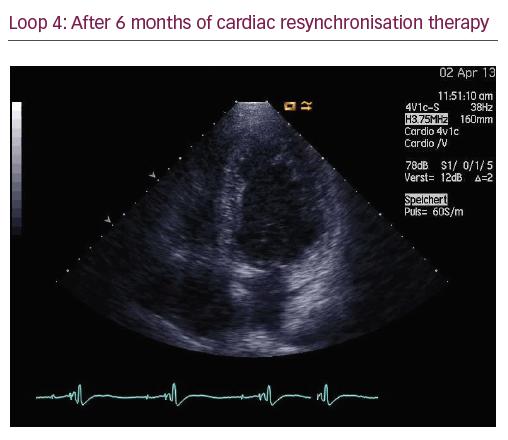
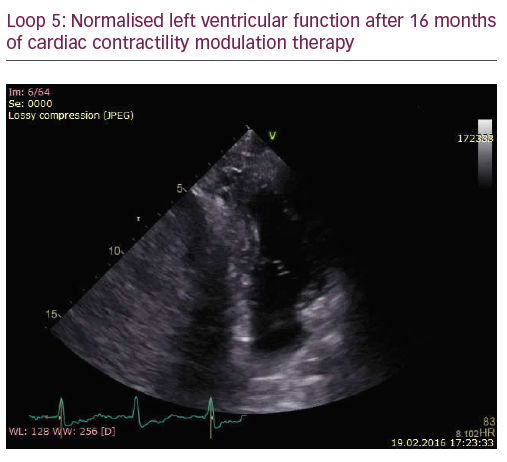
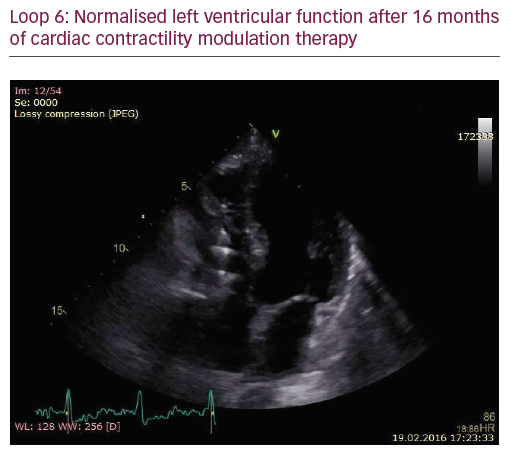
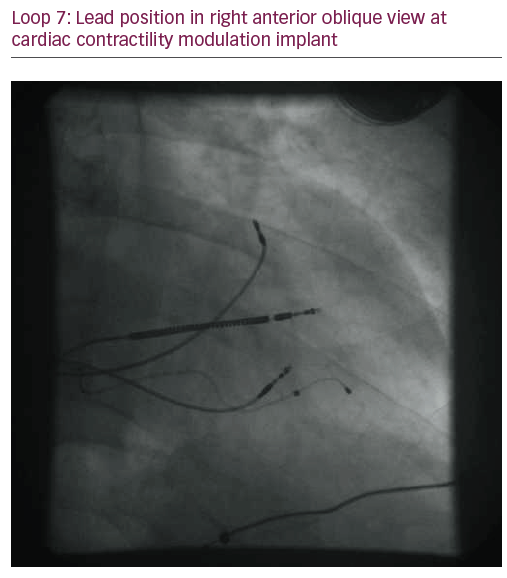
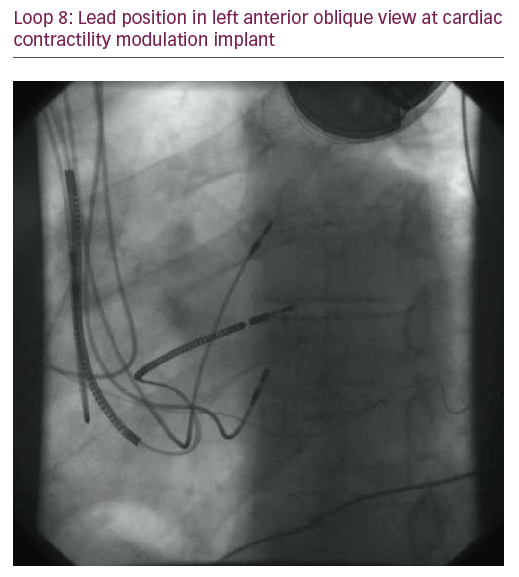
The patient did not respond sufficiently to optimal treatment including CRT despite adequate LV lead position (echocardiogram, Figure 1, Loops 3 and 4). Cardiac contractility modulation (CCM) therapy was initialised by implantation of an Optimizer IV system™ (Impulse Dynamics, Loops 7 and 8, Figure 2). Over a period of 16 months, NYHA class as well as systolic LV function normalised (Figure 1, Loops 5 and 6). Settings for CCM were 30 ms after ventricular sensing, biphasic current of 7.5 V amplitude and 21 ms duration. Of note, the patient continued alcohol abuse.
Discussion
CCM pacing utilises electrical currents to enhance myocardial contractility in heart failure patients. Relatively high (5–7.5 V) voltage impulses are delivered during the absolute ventricular refractory period, after sensing ventricular depolarisation. These currents do not stimulate local ventricular depolarisation, rather they modulate the calcium flux into the myocyte.1 Modulating this calcium flux affects action potential, peak intracellular calcium, increases sarcoplasmic reticulum calcium partly due to influencing phospholamban phosphorylation and altered gene expression.1 These cellular changes may lead to local and remote reverse remodelling.1
The largest prospective randomised trial2 in heart failure patients (NYHA III-IV) with reduced LV ejection fraction (<35%) treated with CCM demonstrated improved quality of life and improved peak VO2 as a measure for exercise tolerance but failed to meet its primary endpoint (change in anaerobic threshold).
Long-term follow-up (>1 year) from prospective randomised trials is lacking for patients treated with CCM. Our case demonstrates improvement in LV function with CCM after 16 months in a patient with insufficient response to CRT. CCM treatment of CRT nonresponders has been described in a series of patients.3 However, previous trials excluded patients with wide QRS or CRT, there is no randomised trial in patients with CRT. This case suggests positive long-term effects of CCM therapy on top of standard therapy for heart failure patients with reduced LV ejection fraction not responsive to CRT.