Background: The learning curve for the novel 3D electroanatomical system Rhythmia is unknown.
Methods: Retrospective data were collected from three UK centres from the introduction of Rhythmia. Procedures considered were de novo and redo atrial fibrillation (AF), cavotricuspid isthmus dependent atrial flutter and left-sided atrial tachycardias. Patients were matched with controls undergoing the same procedure using the well-established Carto3. Assessed metrics were fluoroscopy, radiofrequency ablation and procedure times, acute and long-term success, and complications.
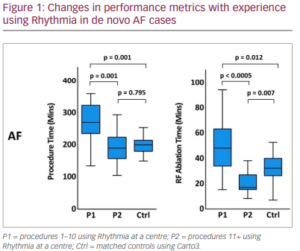
Those procedures that showed correlation with experience using Rhythmia were divided into two groups: P1, where performance markers were improving, and P2, when a plateau had been reached. The threshold number of procedures between P1 and P2 was decided from assessment of scatterplots and comparison with the control group.
Results: A total of 253 study patients with 253 controls were included. Significant correlations existed between procedural efficiency metrics and centre experience for de novo AF ablation (procedure time Spearman’s ρ=-0.624; ablation time ρ=-0.795; both p<0.0005) and de novo atrial flutter (AFlut) ablation (ablation time ρ=-0.566; fluoroscopy time ρ=-0.520; both p=0.001). No such correlations existed for other assessed atrial arrhythmias. For de novo AF and AFlut, metrics significantly improved after 10 procedures in each centre: procedure time (AF only: P1 272.5 ± 65.1 min, P2 192.8 ± 52.5 min; p=0.001 [Figure 1]), ablation time (AF: P1 51.4 ± 22.4 min, P2 20.8 ± 8.8 min; p<0.0005 [Figure 1]; AFlut: P1 15.4 ± 6.9 min, P2 6.2 ± 4.7 min; p<0.0005) and fluoroscopy time (AFlut only: P1 18.1 ± 11.1 min, P2 10.1 ± 9.4 min; p=0.002), and became comparable to controls. Acute success and long-term success did not see significant improvement with experience, but were comparable to the control group throughout. There was no relationship between experience and complications, which were comparable to Carto3 (3.6% in both groups).
Conclusion: A short learning curve was demonstrated with the use of Rhythmia HDx for standardised procedures (de novo AF/AFlut). Procedural performance improved and became comparable to Carto3 following 10 cases at each centre. Clinical outcomes at 6 and 12 months, and complications did not improve with experience and were no different from controls.