Introduction: Severe left ventricular systolic dysfunction (LVSD) is associated with wors e outcomes in heart failure (HF) patients. Persistent atrial fibrillation (AF) is common in this patient group. As part of a nurse-led cardioversion service at a district general hospital we assessed the impact of elective cardioversion (DCCV) for AF in patients with LVSD on their ejection fraction (EF), and compared outcomes in patients selectively pre-treated with Amiodarone for their DCCV to those not.
Methods: A retrospective analysis was undertaken of cardioversions for AF over a 5-year period, recording relevant demographic, medication, rhythm and serial echocardiographic data, and outcomes. Significant LVSD was classified as moderate (ejection fraction [EF] 35–45%) or severe (EF <35%). All patients treated with amiodarone had baseline and serial thyroid, liver, renal function monitored, were counselled on relevant side effects and followed up.
Results: One hundred and three patients with significant LVSD and follow-up echocardiography (TTE) underwent DCCV, with a median age of 66 (IQR 58–73) and mean CHA2DS2-VASc 2.5. The overall mean baseline EF was 30% (SD ± 11), overall follow-up EF (regardless of rhythm at follow-up scan) was 42% (SD ± 12), and the mean delta EF + 12% improvement (SD ± 11). At follow-up TTE, 66% (68/103) of patients were in sinus rhythm (SR) and 34% (35/103) in AF. Sixty-two out of 68 (91%) patients in SR at follow-up TTE were also treated with HF medications, versus 33/35 (94%) of those in AF. Sixty-one out of 68 (90%) of patients in SR at follow-up TTE had any improvement in EF versus 21/35 (60%) of patients in AF (p=0.0007). For patients in SR, the mean baseline EF was 31% (SD ± 10) and mean follow-up EF 47% (SD ± 9), versus the AF at follow-up scan patients’ mean baseline EF 27% (SD ± 12) and mean follow-up EF 35% (SD ± 13). The mean delta EF of patients still in SR at follow-up scan was 15% (SD ± 10) versus 8% (SD ± 11) for patients who had reverted to AF (p=0.0004). Table 1 summarises outcomes in patients pre-treated with amiodarone versus those not, for cases with available follow-up TTE data.
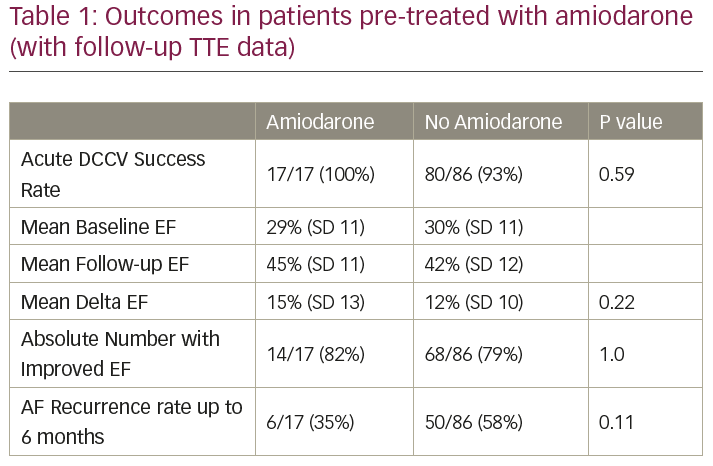
A separate analysis of our data-set included patients currently awaiting a post-DCCV TTE demonstrated a statistically significant improvement in 6-month AF recurrence rate.
pre-treated with Amiodarone. Further study into long-term rhythm control and ablation outcomes is needed.
Conclusion: Restoration of SR in a cohort of patients with AF, severe LVSD, who are on good medical therapy significantly improves left ventricular EF. This reinforces the importance of maintenance of SR for HF patients. There was also a trend towards improved medium term outcomes in patients.







