Background: Data on quantitative assessment of dynamic ST changes during 12-lead 24-hour ambulatory ECG in Brugada Syndrome (BrS) are limited.
Aim: To investigate whether the quantitative analysis of ST changes during 12-lead 24-hour ambulatory ECG could contribute to the diagnosis and risk stratification of BrS.
Methods: A total of 147 BrS patients (55% male, mean age 47±15 years, 10 with spontaneous diagnostic type 1 pattern, 22 with arrhythmic events – syncope, aborted cardiac arrest or appropriate ICD intervention) and 68 healthy subjects (55% males, mean age 29±11 years) with no previous cardiac symptoms, no family history of sudden death and normal cardiac investigations, were included in the study. Digital 12-lead 24-hour ambulatory ECG were recorded. One-minute interval averaged values of the ST segment in lead V1-V2 positioned in the 4th, 3rd and 2nd intercostal spaces were obtained and plotted over time. Mixed-effects models were applied to the continuous longitudinal ECG data (log-transformed to account for the inter-individual and intra-individual variability) to evaluate their association with: 1) the diagnosis of BrS; 2) the response to sodium channel blocker challenge; 3) the presence of a diagnostic type 1 during the recording; 4) the presence of arrhythmic events.
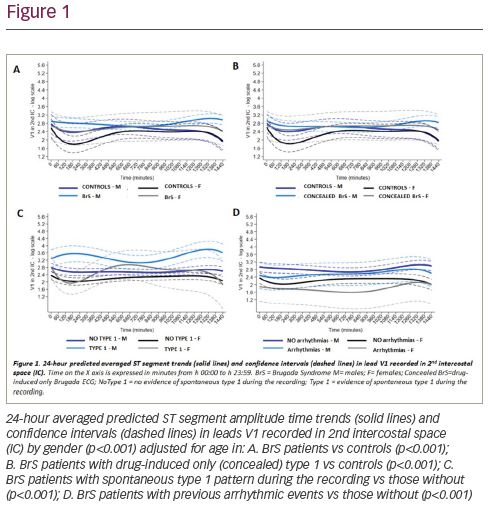
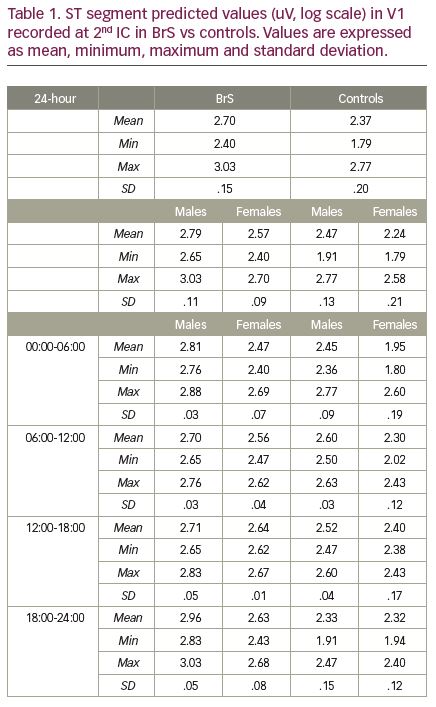
Results: There were statistically significant differences in analysed 24-hour ST trends for: A) all subjects with a diagnosis of BrS vs controls; B) BrS patients with a drug-induced type 1 pattern only versus controls; C) BrS subjects with a spontaneous type 1 pattern during the recording versus those without; D) BrS with arrhythmic events versus those without. Average 24-hour trends showed different patterns across gender and clinical diagnosis of BrS (p<0.001) adjusted for age (Figure 1). Table 1 shows the predicted ST segment amplitude averages obtained from the model in lead V1 recorded at 2nd intercostal space in BrS patients vs controls, also stratified by gender and four time segments during the 24-hour cycle.
Conclusions: The quantitative analysis of ST changes during 24-hour ambulatory ECG may be useful for the identification of subjects with a diagnosis of BrS, those with a spontaneous type 1 pattern and/or arrhythmic events.