The current pandemic of coronavirus disease 2019 (COVID-19) is an unprecedented public health emergency, with worldwide health, social and economic implications. In contrast to other modern epidemics, little prior knowledge of the pathogen existed. Whilst this initially hampered both patient management and the public health response, we have observed a remarkable worldwide attempt to provide information to guide both clinicians and governments, with 11,478 individual publications registered in the World Health Organisation database of COVID-19 research, as of May 2020.1
Early data on COVID-19 suggested poor outcomes amongst those with established cardiovascular disease and risk factors. Case reports have described patients presenting with myopericarditis and apparent acute coronary syndromes, and angiotensin-converting enzyme 2 (ACE-2) was identified as the virus’s portal of entry into host cells; these observations highlight the need for research into the cardiovascular aspects of the disease.2 Whilst a substantial proportion of the published material, in this field to date, consists of opinion pieces and narrative reviews, a number of key findings have emerged and have been summarised below.
Implications of pre-existing cardiovascular disease in COVID-19
By current estimation, the case fatality rate for COVID-19 is 1.5–3.0%, and the infection fatality rate is around 0.7%, although this is not evenly distributed throughout the population.3 Drs Wu and McGoogan, from the Chinese Centre for Disease Control and Prevention in Beijing, reported markedly higher fatality rates in patients with cardiovascular disease (10.5%), diabetes (7.3%) and hypertension (6.0%) in 72,314 cases from China.4 These findings were replicated in the Italian population.5 Closer inspection, however, revealed the prevalence of these risk factors is largely in keeping with the population prevalence of these comorbidities when analysed by age. This was also apparent in the loss of an independent effect on mortality in multivariate analyses.4
Myocardial injury and COVID-19
Zhou and colleagues first described the clinical course and predictors of outcome in 195 consecutive patients who received inpatient care for COVID-19 in Wuhan, China.6 One hundred and thirty-seven survived and 54 died, with 26% requiring intensive care unit (ICU) admission. Complete data were available for 171 patients and were used to build a regression model predicting outcomes. There are several interesting cardiovascular aspects.
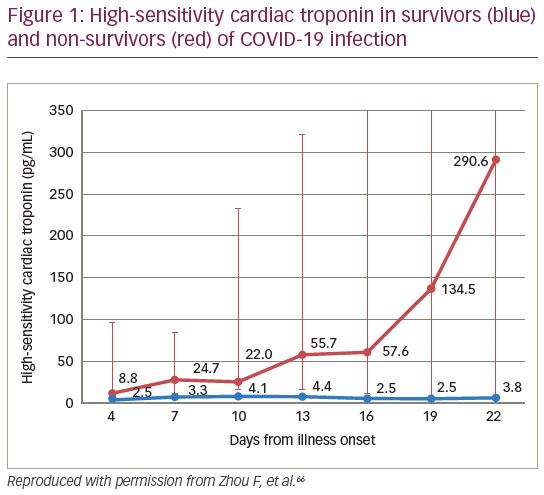
Acute cardiac injury (high-sensitivity cardiac troponin I [hs-cTnI] >99th centile, 28 pg/mL in this series) was common in patients who did not survive (59% of non-survivors versus 1% of survivors). In those who died, there was a steady upwards trend in troponin from the day of admission, though the confidence intervals (CIs) were extremely wide (Figure 1). However, it should be noted that the non-survivors were largely the critically ill ICU population, whilst the survivors were mainly cared for outside of critical care. cTnI levels were effective predictors of mortality on univariate analysis, but on multivariate analysis, the prognostic value of cTnI was lost; only age, Sequential Organ Failure Assessment (SOFA) score, and d-dimer on admission remained predictive of mortality.
A meta-analysis of 2,389 patients from 13 studies was also published in the American Journal of Emergency Medicine.7 A hs-cTnl >99th percentile was considered to represent cardiac injury. The primary outcome was mortality and secondary outcomes were acute respiratory distress syndrome (ARDS), the need for ICU care and severe COVID-19 (any respiratory distress, saturated oxygen ≤93%, partial pressure of arterial oxygen/fractional inspired oxygen [PaO2/FiO2] ≤300 mmHg, or a critical complication). High-sensitivity-cTnI was higher in patients with primary and secondary outcomes (mean difference 10.38 pg/mL [95% CI 4.44–16.32]). Limitations include a high degree of heterogeneity in the studies included, evidence of publication bias, and that it is difficult to know whether there was overlap in patient cohorts between studies.
Finally, a retrospective case-control study compared two independent cohorts of 73 patients with COVID pneumonitis admitted over a 7-week period in Wuhan, and 75 patients with influenza A (H1N1) ARDS admitted over a 30-month period in Beijing.8 The H1N1 group were sicker (SOFA score 5 [interquartile range 4–8] versus 2 [2–4] p<0.001; PaO2/FiO2 <100 mmHg in 48% versus 7%, p<0.001) and were less likely to have a history of cardiovascular disease than the group with COVID (11% versus 32%, respectively, p=0.002). Peak troponin I levels were higher in the H1N1 group (140 ng/L [interquartile range [20–370] versus 30 ng/L [30–50], respectively, p=0.014) but this was no longer significant when adjusted for other factors.
Interpretation
Whilst the groups in the aforementioned studies are different and this limits the certainty with which we can draw conclusions, it is likely that, whilst cardiac troponin is a prognostic marker in COVID-19, there is no evidence it provides incremental prognostic information in relation to other markers of disease severity. Furthermore, whilst there have been descriptions of individual patients presenting with overt cardiovascular pathology, the majority of even critically ill patients with COVID-19 have modest degrees of myocardial injury reflecting their overall illness severity, rather than specific cardiac involvement contributing to mortality.
Imaging in COVID-19
The role of echocardiography in COVID-19 has been uncertain, balancing the importance of assessing critically ill patients (in whom the incidence of cardiac complications is unclear) against unnecessary exposure of team members. However, a study of 120 patients from China shows the echocardiographic assessment of right ventricular (RV) function, and strain in particular, to be useful in this population.9 Several parameters of RV function – RV longitudinal strain (RVLS), RV fractional area change and triscuspid annular plane systolic excursion (TAPSE) – alongside male gender and the presence of ARDS were found to predict mortality, which occurred in 18 patients (15%). RVLS and TAPSE retained their prognostic value in multivariate modelling, with RVLS providing the optimum prognostic value. Whether the observed RV dysfunction represents direct myocardial dysfunction, pulmonary thromboembolic disease or the severity of ARDS, remains to be seen.
Angiotensin-converting enzyme inhibitors and angiotensin receptor blockers in COVID-19
A recent review published in the New England Journal of Medicine elegantly summarised the background to this issue.10 Concerns were initially raised based on select preclinical studies, stating that ACE inhibitors and angiotensin receptor blockers (ARBs) may increase ACE2 expression and provide a more ready portal of entry for COVID-19 in patients being treated with these drugs. However, little reliable data existed and none specific to COVID-19, whilst abrupt withdrawal of these drugs in high-risk patients is likely to be harmful. A clear consensus emerged across international cardiac and hypertension societies that these medications should be continued.
Subsequently, Zhang and colleagues published a retrospective
nine-centre study of 1,128 patients with hypertension diagnosed with COVID-19 (188 patients on ACE inhibitors/ARBs) in Hubei Province, China from 31 December 2019 to 20 February 2020.11 Unadjusted mortality was lower in the group on ACE inhibitors/ARBs compared with those who were not (3.7% versus 9.8%, p=0.01). There were baseline differences between the two groups (more coronary artery disease, diabetes and renal disease in the ACE inhibitor/ARB group and more stroke in the other). The authors carried out a variety of techniques to account for these differences (including an adjusted mixed-effect Cox model and propensity score-matched analyses) and concluded that the mortality benefit associated with prior ACE inhibitors/ARBs remains.
Interpretation
Notwithstanding limitations, it can probably be concluded that ACE inhibitor/ARB use is not associated with worse outcomes. The European Society of Cardiology and other international guidelines recommend continuing ACE inhibitors and ARBs in patients with COVID-19 and also in our general hypertensive and heart failure populations, for now. Randomised trials investigating the use of losartan in COVID-19 are ongoing (see ClinicalTrials.gov).
Endothelial cell infection in COVID-19
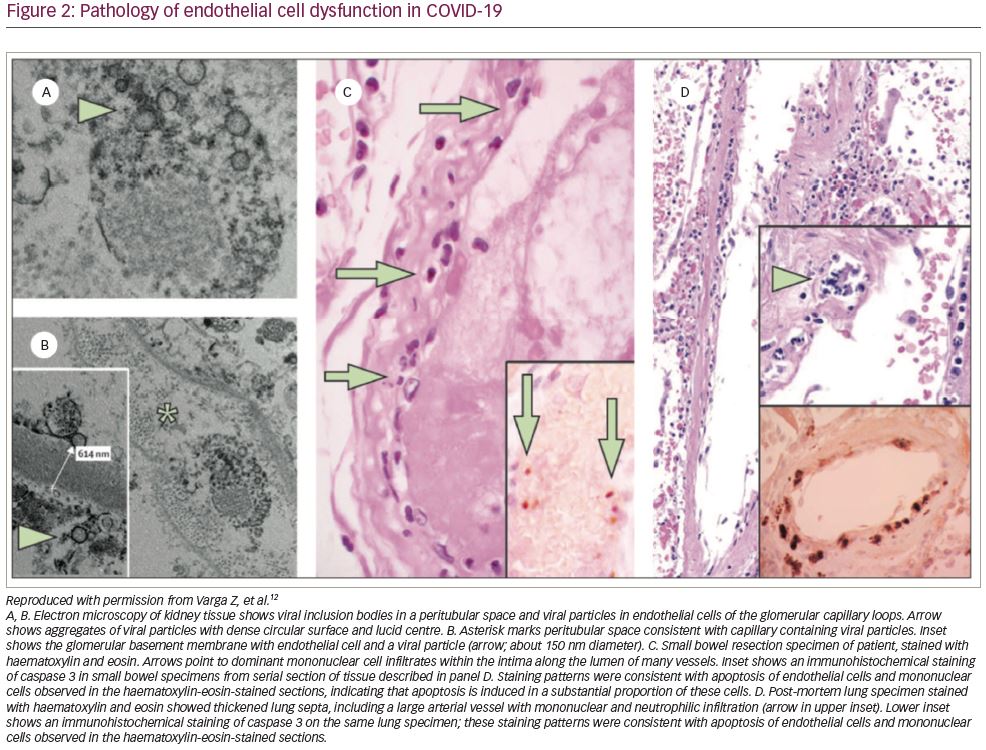
A research letter, published in The Lancet, described histological findings in three patients with COVID-19: a renal transplant recipient with coronary artery disease and hypertension who died of multiorgan failure; a female with diabetes and hypertension who died of multiorgan failure including ST-elevation myocardial infarction (STEMI), RV failure and bowel ischaemia; and a man with hypertension who had small bowel resection for ischaemia and survived.12 They found histological evidence of endothelial inflammation in the lungs, heart and submucosal vessels of the small bowel. Electron microscopy demonstrated viral inclusion in the endothelial cells of a transplanted kidney of one patient (Figure 2A and B).
Interpretation
It is unclear whether endotheliitis is a direct consequence of endothelial cell infection, which may explain the relation with factors associated with endothelial dysfunction. Future post-mortem series would be instructive.
Acute coronary syndromes in COVID-19
There has been significant concern regarding patients with COVID-19 and acute coronary syndrome-like presentations, some of which have appeared to be true acute myocardial infarctions and others a number of ‘STEMI mimics’. A case series of 18 patients with COVID-19 presenting as possible STEMI to New York hospitals, was published in the New England Journal of Medicine.13 Ten out of 18 patients were labelled as having ‘noncoronary myocardial injury’, of whom, 90% died, compared with 50% of those with true acute myocardial infarction. Some of the most interesting data are in the supplementary appendix, including all 18 electrocardiograms (ECGs), which prove interesting to review. Half the whole cohort had normal left ventricular ejection fraction on echocardiogram. There were substantial limitations, however – only half of all patients had angiography, of whom six had percutaneous coronary intervention (PCI).
Another major concern has been a reduction in the number of patients presenting with symptoms of acute coronary syndromes. A study analysing data from nine ‘high-volume’ centres across several states in the USA (defined as performing ≥100 primary PCI procedures per year) demonstrated a 38% reduction in such presentations in March 2020.14 This is consistent with the 40% reduction previously reported from Spain. Speculated reasons for falling numbers of patients with STEMI included avoidance of medical care due to concerns of contracting COVID-19, misdiagnosis as suspected COVID-19, or a shift to conservative care.14
Interpretation
Whilst diagnostically challenging, the outcomes of patients with COVID-19 and suspected acute myocardial infarction are very poor, including in those with a final diagnosis of STEMI (likely because of delayed/no PCI). It is important to make a full and quick assessment (ECG, echocardiogram) of these patients and not delay angiography if felt appropriate. Simultaneously, there may be a substantial number of patients without COVID who have acute coronary syndromes who may not present acutely; whether this results in an increase in presentations with cardiogenic shock or acute heart failure remains to be seen.
Review and guidance
On 21 April 2020, the European Society of Cardiology published a document on understanding and managing cardiovascular complications of COVID-19, from pathophysiological insights to personal protective equipment. This is a pragmatic document and a good resource.15
Conclusions
The pandemic has been remarkable for the rapid dissemination of data on cardiovascular involvement. Though the presence of, and risk factors for, cardiovascular disease are associated with adverse outcomes in COVID-19, the current evidence points towards these being surrogate markers for initial poor health and acute disease severity, rather than direct cardiac involvement in the majority of cases. Drugs affecting the renin–angiotensin–aldosterone system appear safe and possibly protective (though more data is needed), and the role of the vascular endothelium in the pathophysiology of COVID-19 merits investigation. The small proportion of patients who do have evidence of direct cardiac involvement merit early assessment and, if appropriate, intervention by specialists in cardiovascular medicine.