Background: Cardiac implantable electronic device (CIED) infection has risen faster than the volume of procedures. Many measures against CIED infection have been tried, but only peri-operative antibiotics and an antibiotic-eluting envelope have proved effective. Taurolidine is a long-established antimicrobial agent with a wide range of chemical activities and biological effects. The effectiveness of a taurolidine-containing solution in preventing CIED infection was assessed in this observational study.
Methods: All the hardware (leads, suture sleeves, pulse generator) was washed and the device pocket irrigated with an adjunct antimicrobial solution, which could be 3% hydrogen peroxide (H2O2), taurolidine in a galenic formulation or TauroPace™ (TP, Tauropharm, Bavaria, Germany), during any invasive procedure (e.g. generator replacement) involving a CIED system at the authors’ institute. Before 01/01/2020, the choice of antimicrobial solution was at the operator’s discretion. Afterwards, only TP was used. All CIED procedures performed at the author’s institute between 01/01/2017 and 28/02/2022 were included for analysis. Patients who received the galenic taurolidine formulation were excluded from analysis. The primary endpoint was CIED infection according to the Novel 2019 international diagnostic criteria. The secondary endpoint was any serious adverse events (SAE, e.g. pneumothorax) possibly related to the use of the antimicrobial solution, the CIED or the procedure, and all-cause mortality. The follow-up duration was standardised to 3 months as only acute and sub-acute infection post-CIED procedure was of interest. The procedures and not the patients were the data units. Patients who underwent more than one procedure were deemed to have been censored for the initial treatment group and re-classified as new data units (with or without cross-over to the other treatment group) at the time of the second procedure.
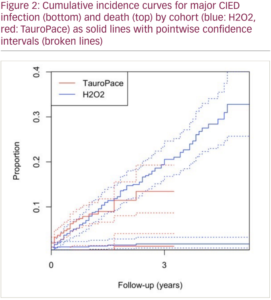
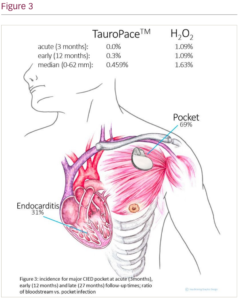
Results: Between January 2017 and February 2022, 654 procedures were conducted with adjunct TP in 631 distinct patients, and 551 procedures with adjunct H2O2 in 532 distinct patients (Figure 1). The TP group had significantly more host risk factors for infection (e.g. inexperienced operator) than the H2O2 group (p=0.0017), but similar device- and procedure-specific risk factors (p=0.22). Within 3 months post-index procedure, CIED infection occurred in 0/654 (0.0%) in the TP group and 6/551 (1.09%) in the H2O2 group (95% CI 0.5% to 2.36%; p=0.0075) (Figure 2, Figure 3). Death occurred in 23/654 (3.5%) of the TP group and 14/551 (2.5%) in the H2O2 group (95% CI -2.98% to 1.05%; p=0.32). Non-infection-related SAEs were rarer in the TP group than in the H2O2 group (95% CI -4.79% to 0.26%; p=0.0802) (3.8% vs 6.0%). The hazard ratio for major pocket infection was 0.41 (0.11 to 1.56) without adjustment and 0.35 (0.09 to 1.37) when adjusted for number of patient risk factors in a Cox regression using all follow-up (median 15 months).
Conclusions: The use of TP as an antimicrobial solution during CIED procedures may have been a cause of a lower rate of acute and delayed CIED infection compared with 3% H2O2 in an observational study. The clinical utility of TP in reducing CIED infection will need to be assessed in a randomised clinical trial.