Background: The WiSE-CRT system delivers wireless endocardial pacing to achieve cardiac resynchronisation therapy. It consists of a subcutaneous transmitter, battery and endocardial electrode. The transmitter detects right ventricular pacing from the co-implant and delivers ultrasound energy to the electrode causing left ventricular (LV) pacing and cardiac resynchronisation. Ideally, the electrode should be placed in an area of late contracting or activating areas whilst avoiding myocardial scar. Furthermore, the transmitter must be placed in close proximity to the electrode but since this is implanted before the electrode, identifying the optimal location pre-procedurally is important. Currently, the optimal location for both the transmitter and electrode is unknown. We hypothesised that pre-procedural imaging to estimate scar and measure mechanics combined with modelling and simulation to predict activations times would be able to predict the optimal location for both the electrode and transmitter.
Objectives: To determine whether integrated cardiac computed tomographic (CT) imaging and simulation workflow can successfully guide placement of the transmitter and electrode.
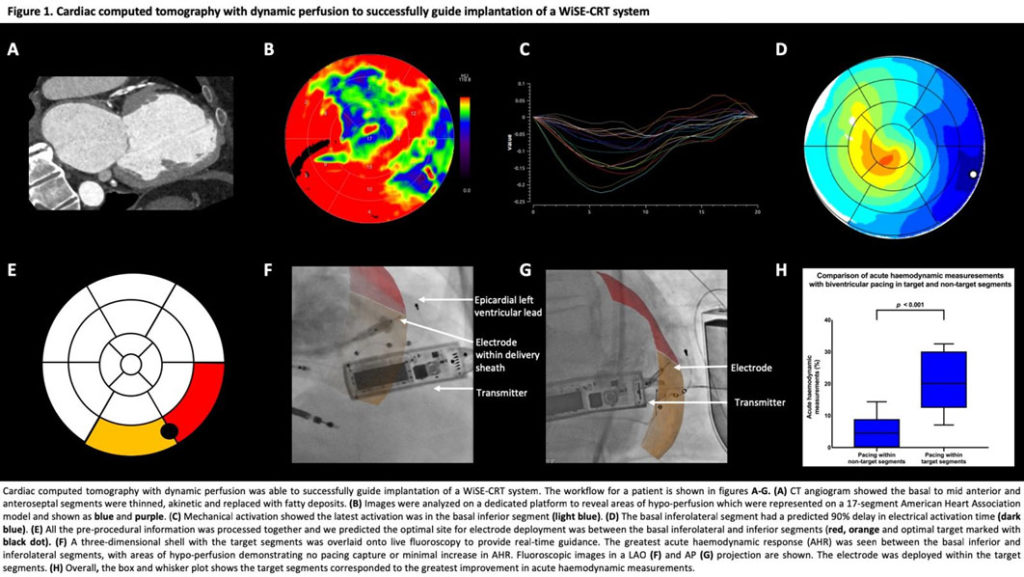
Methods: Patients eligible for the WiSE-CRT system were recruited. They underwent a cardiac CT with stress perfusion to identify areas of perfusion heterogeneity and a retrospective gated CT to measure latest mechanical activation and patient specific modelling to predict electrical activation. These were analysed on a dedicated platform to identify the target segments for electrode deployment. We modelled the power coverage for the transmitter and identified the optimal location for this. The pre-procedural target segments for electrode deployment were merged and overlaid onto live fluoroscopy to help guide the implant procedure. A pressure wire in the LV cavity was used to measure the acute haemodynamic response (AHR) with biventricular endocardial pacing in different myocardial segments. Segments with an improvement of ≥10% in dP/dt(max) were considered a favourable location for the LV electrode based on our previous AHR studies.
Results: Five consecutive patients were studied with a mean age of 70.3 ± 6.9 years; NYHA Functional class 2.8 ± 0.4; QRS duration 166.6 ± 34.5ms; LVEF 28.4 ± 6.2%. Two patients had ischaemic cardiomyopathy and three non-ischaemic cardiomyopathies. The transmitter was modelled pre-procedurally and placed in the recommended site in all cases. Overall, the AHR of pacing within 24 different LV myocardial segments was tested; a significantly greater number of target segments had an AHR ≥10% versus non-target segments (87.5 versus 18.75%, p=0.002). Furthermore, all segments with a perfusion defect on stress CT had an AHR less than 10%. Therefore, the electrode was deployed within the target segment in all cases.
Conclusion: We successfully used cardiac computed tomography and simulation to guide the optimal placement for the WiSE-CRT transmitter and electrode.