Drug-eluting technology is a widely accepted, proven treatment for coronary artery stenosis and occlusions, with superior results when compared with bare metal stents or plain old balloon angioplasty (POBA).1,2 Many studies also proved drug-eluting stents (DES) and drug-coated balloons (DCB) to be superior to POBA when treating lower limb arteries.3,4
Paclitaxel is a cytostatic chemotherapy drug belonging to the taxane group, and is a commonly used systemic treatment for several carcinomas, including lung, breast and prostate. When treating peripheral arterial disease, some current devices contain paclitaxel to prevent restenosis.5 The drug is applied directly to the vessel wall, where it acts locally to prevent over-response and cell proliferation (intimal hyperplasia) triggered after an injury to the vessel wall, such as stretching in balloon angioplasty and/or stent deployment.
After more than a decade on the market, the reputation, and therefore use, of DES and DCB containing paclitaxel for peripheral arterial disease suffered a massive setback when, in December 2018, a meta-analysis of 28 randomized controlled trials with 4,663 patients showed an increase in all-cause death in patients receiving paclitaxel, compared with control groups. Katsanos et al. reported a significant increase in all‐cause death at 2 years with paclitaxel versus controls (7.2% versus 3.8% crude risk of death, respectively; risk ratio, 1.68; 95% confidence interval [CI], 1.15–2.47; number‐needed‐to‐harm, 29 patients [95% CI, 19–59]). All‐cause death up to 5 years (three randomized controlled trials with 863 cases) further increased with paclitaxel versus controls (14.7% versus 8.1% crude risk of death, respectively; risk ratio, 1.93; 95% CI, 1.27–2.93; number‐needed‐to‐harm, 14 patients [95% CI, 9–32]).6
Following this publication, interventionalists and healthcare officials attempted to explain this phenomenon and debated how to fill the gap left by this relevant therapy until further studies were completed to prove or disprove the conclusions. As a result, the indication for paclitaxel-eluting devices has narrowed; it is now mostly restricted to lesions with high risk of restenosis, such as in-stent stenosis.7,8 Physicians are instructed to weigh up the risks and benefits of using paclitaxel-eluting devices, and to fully convey risks to the patient within a well-informed, balanced and documented informed consent process.
While waiting for some long-term data on paclitaxel to be published, the focus has slowly shifted to some promising, mainly limus-based drugs, such as sirolimus. In this article, we briefly summarize available evidence on drug-eluting technology for treating peripheral arterial disease.
The focus of the studies included in this article was to evaluate the treatment with DCBs on symptomatic patients with a Rutherford score of 2 or higher. Some studies also focused on comparing devices directly. All trials discussed studied patients with claudication. Endpoints were usually primary patency, as assessed via ultrasound, and freedom from target lesion revascularization. Specific lesion characteristics, such as length and severity, vary between the studies, although the lesions were mainly located on the superficial femoral artery or the upper popliteal artery.
IN.PACT SFA randomized trial
The IN.PACT SFA trial (Randomized Trial of IN.PACT Admiral® Drug Coated Balloon vs Standard PTA for the Treatment of SFA and Proximal Popliteal Arterial Disease; ClinicalTrials.gov identifier: NCT01175850) enrolled 331 patients with intermittent claudication or ischaemic rest pain attributable to SFA and PPA disease, randomized in a 2:1 ratio for treatment either with the paclitaxel-coated IN.PACT Admiral DCB (Medtronic, Dublin, Ireland) or standard percutaneous transluminal angioplasty (PTA) of the SFA and/or PPA. Mean lesion length and the percentage of total occlusions for the DCB and PTA arms were 8.94 ± 4.89 and 8.81 ± 5.12 cm (p=0.82) and 25.8% and 19.5% (p=0.22), respectively.14
Three-year data showed a higher patency and freedom from target lesion revascularization rate for the DCB group, namely 69.5% patency for DCB versus 45.1% for the POBA, and 84.8% freedom from target lesion revascularization for the DCB versus 68.9% for the POBA. The authors claim there were no device- or procedure-related deaths, adjudicated by an independent Clinical Events Committee.15
Authors from the IN.PACT trial analysed two single-arm and two randomized trials of paclitaxel-coated balloons, with 1,837 patients, for all-cause mortality. The results after stratification showed no correlation between exposure to different doses of paclitaxel and mortality. In addition, when comparing with POBA, no statistically significant increase in mortality was detected (p=0.092).16
COMPARE and RANGER II SFA trials
The COMPARE trial (ClinicalTrials.gov identifier: NCT02701543) compared two DCBs: the Ranger™ DCB (Hemoteq, Würselen, Germany) and the IN.PACT™ Admiral™ DCB or IN.PACT Pacific™ DCB (Medtronic Vascular, Santa Clara, CA, USA).9,10 This trial compared devices in a 1:1 ratio in 414 patients with peripheral arterial disease. The primary aim of the trial was to prove that the low-dose Ranger DCB is non-inferior to the higher dose IN.PACT Admiral/Pacific DCB. The Ranger DCB is a paclitaxel-coated balloon with a drug density of 2 µg/mm², whereas the IN.PACT Admiral/Pacific DCBs have a drug density of 3.5 µg/mm².10
Twelve-month results were published in July 2020, concluding that the Ranger DCB is non-inferior to the IN.PACT Admiral/Pacific DCBs, with a 1-year event-free survival of 88.4% for the Ranger DCB versus 89.4% for the IN.PACT DCBs.10 Bailout stenting rates were 25–30% for both groups, mainly when treating longer (>20 cm) lesions. This ongoing study will follow patients for up to 2 years for patency and up to 5 years for target lesion revascularization.10
The Ranger II SFA trial (RANGER™ Paclitaxel Coated Balloon vs Standard Balloon Angioplasty; ClinicalTrials.gov identifier: NCT03064126) is ongoing.11 The preliminary 12-month follow-up results of 376 symptomatic (Rutherford 2–4) patients presenting stenosis up to 180 mm long and/or occlusions up to 100 mm presented a freedom from target lesion revascularization rate of 94.5% for the Ranger DCB and 83.5% for the standard, uncoated balloon (3:1 randomization).12 These results are in line with previous studies involving the Ranger DCB.13
ILLUMENATE trials
The ILLUMENATE EU (CVI Drug Coated Balloon European Randomized Clinical Trial; ClinicalTrials.gov identifier: NCT01858363) and ILLUMENATE Pivotal (Pivotal Trial of a Novel Paclitaxel-Coated Percutaneous Angioplasty Balloon; ClinicalTrials.gov identifier: NCT01858428) randomized controlled trials together included 594 (n=294 ILLUMENATE EU, n=300 ILLUMENATE Pivotal) patients (Rutherford class 2–4) who received either the low-dose, paclitaxel-coated Stellarex DCB (Spectranetics Corp., Colorado Springs, CO, USA) or POBA in a 3:1 ratio. Mean lesion length in the ILLUMENATE EU was 72 mm and 71 mm, and 19.2% and 19.0% of lesions represented total occlusions, respectively. In the ILLUMENATE Pivotal, baseline characteristics were similar between groups, the mean lesion length was 83 mm, and 44.0% were severely calcified.17,18
In the ILLUMENATE EU trial, the primary patency at 2 years was higher in the DCB group than the POBA group (75.9% versus 61.0%, respectively; p=0.025), and the rate of clinically driven target lesion revascularization (CD-TLR) was significantly lower in the DCB group than the POBA group (12.1% versus 30.5%, respectively; p<0.001).17 There were no limb amputations in either group, and all-cause mortality was similar between groups (6.5% DCB versus 5.1% POBA; p=1.00).19
In the ILLUMENATE Pivotal Study, the primary patency rate was significantly higher with DCB (76.3% for DCB versus 57.6% for PTA, p=0.003). Primary patency per Kaplan-Meier estimates at day 365 was 82.3% for DCB versus 70.9% for PTA (P=0.002). The rate of clinically driven target lesion revascularization was significantly lower in the DCB cohort (7.9% versus 16.8%, P=0.023). Improvements in ankle-brachial index, Rutherford class, and quality of life were comparable, but the PTA cohort required twice as many revascularizations.18
The 5-year data presented at the Leipzig Interventional Course 2021 (LINC, 25–29th January 2021, online) demonstrated no statistical difference in the mortality; the ILLUMENATE EU trial showed 19.3% mortality in the DCB group versus 19.4% in the POBA group, and the ILLUMENATE Pivotal randomized controlled trial showed 21.2% mortality in the DCB group versus 20.2% in the POBA group.20
SELUTION SFA trial
The SELUTION SLR™ DCB (M.A. Med Alliance SA, Nyon, Switzerland) is a sirolimus-eluting balloon. Of the 50 patients with symptomatic moderate-to-severe lower limb ischaemia (Rutherford categories 2 or 3) enrolled on the SELUTION SFA trial (ClinicalTrials.gov identifier: NCT02941224) between October 2016 and May 2017 to four centres in Germany; 42 patients completed the 24-month follow-up.21
The primary trial objective was comparison of angiographic late lumen loss at 6 months against an objective performance criterion (OPC) value of 1.04 mm for uncoated balloon angioplasty. Secondary endpoints included device, procedural, and clinical success; clinical and imaging assessments of primary patency and restenosis; functional assessments including Rutherford category and ankle-brachial index (ABI); and major adverse events (composite of cardiovascular mortality, index limb amputation, target limb thrombosis and clinically-driven target lesion revascularization [CD-TLR]).
The currently available published data is on the 6-month outcome. The rate of primary patency by duplex ultrasound was 88.4%, and freedom from angiographic binary restenosis was 91.2%. Through 6 months, there was significant improvement over baseline in Rutherford categories (p<0.001) and in ABI measurements (p<0.001). A single case (2.0%) of CD-TLR occurred at 5 months. There were no other major adverse events.22 According to the presentation held at the 2019 Vascular Interventional Advances (VIVA) Annual Conference in Las Vegas, the freedom from target lesion revascularization rate was 87.5% after 1 year, and all-cause mortality was 0.0%.23
Drug-eluting stents
Long and complex lesions with balloon angioplasty alone are prone to increased rates of flow-limiting dissection and recoil, with higher incidence of restenosis and re-occlusions in the years following the index procedure.24 Bailout or primary stenting of these types of lesions have been consistently proven more effective.25,26
Many studies and meta-analyses have shown that using DES results in decreased rates of restenosis and target lesion revascularization when compared with bare metal stents. For instance, studies on the paclitaxel-coated Zilver® PTX® drug-eluting peripheral stent (Cook Medical, Bloomington, IN, USA) presented promising results, even for patients with Trans-Atlantic Inter-Society Consensus Document II (TASC II) type C or D lesions, which produce worse patient outcomes.27
Early studies with stents coated with drugs other than paclitaxel have, in turn, failed to provide good results. The 2006 SIROCCO trial (A Study of the SMART Stent in the Treatment SFA Disease; ClinicalTrials.gov identifier: NCT00232869) showed similar results between the sirolimus-eluting S.M.A.R.T.® Vascular Stent (Cordis Corporation, Miami Lakes, FL, USA) and the uncoated version of the stent.28 After 24 months, the restenosis rate in the sirolimus group was 22.9% versus 21.1% in the bare metal stent group (p>0.05). Although these results were as expected, the trial showed no relevant benefits of DES over the bare metal stent.29
The single-arm, non-randomized STRIDES trial (A Safety and Efficacy Study of the Dynalink®-E Everolimus Eluting Peripheral Stent System; ClinicalTrials.gov identifier: NCT00475566; 2011), with 104 participants, analysed an everolimus-eluting, self-expanding nitinol stent.30 The results suggested improved primary patency at 6 months, although this improvement was not sustained at 12 months.31
Zilver PTX trials
The Zilver PTX stent consists of a self-expanding nitinol stent with a polymer-free, paclitaxel coating. It was among the first paclitaxel-eluting stents approved and marketed (first FDA submission 2001, CE mark approval 2009).32,33
The initial analysis of the Zilver PTX stent was divided into two trials. The first was a randomized trial (ClinicalTrials.gov identifier: NCT00120406)comparing the Zilver PTX stent with a control group consisting of PTA and, should a bailout stent be required, a possible second randomization between a plain bare metal stent and the Zilver PTX stent.34,35 The second trial was single arm, designed to test the stent on a real-world scenario, with no limits on the length of the lesions and the underlying reason for stenting, including in-stent restenosis.36
The randomized trial enrolled patients with symptomatic femoropopliteal artery disease (Rutherford category 2–6); approximately 91.0% had claudication; 9.0% had critical limb ischaemia.35 Patients were randomly assigned to DES (n=236) or PTA (n=238) groups. Patients experiencing acute PTA failure underwent secondary randomization to provisional bare metal stent (n=59) or DES (n=61). The maximal lesion length was 70 mm in the first phase and 140 mm in the second phase of this trial. The mean lesion length was 54 ± 41 mm for the Zilver PTX stent group and 53 ± 40 mm for the control group. Occlusions were found in 30.0% and 25.0% of the patients in the DES and control groups, respectively. In total, 546 stents were implanted; 453 were Zilver PTX stents and 93 were plain bare metal stents. The primary patency at 5 years was 66.4% for the DES group and 43.4% in the control group. Freedom from target lesion vascularization was 83.1% versus 67.6% in the DES and control groups, respectively, again favouring the DES over the control group. Worth noting is the higher 5-year all-cause mortality rate of 16.9% for the DES group versus 10.2% for the control group (p=0.03).34
The single-arm study enrolled 787 patients (Rutherford category 2–6) undergoing DES treatment. A total of 900 lesions (24.3% restenotic lesions, of which 59.4% were in-stent stenoses) were treated with 1,722 Zilver PTX stents; the mean lesion length was 99.5 ± 82.1 mm. The 12-month data included an 89.0% event-free survival rate, an 86.2% primary patency rate and a 90.5% rate of freedom from target lesion revascularization.36
IMPERIAL trial
Following the initial MAJESTIC trial (ClinicalTrials.gov identifier: NCT01820637) on Boston Scientific’s Eluvia™ drug-eluting vascular stent system (paclitaxel, polymer-coated; Boston Scientific, Maple Grove, MN, USA), which presented a freedom from target lesion revascularization of 85.3% at 3-year follow-up (index n=57), the IMPERIAL trial (ClinicalTrials.gov identifier: NCT02574481) compared the Eluvia stent with the Zilver PTX stent.37–39
In this single-blind, non-inferiority study, 465 patients were randomly assigned 2:1 to receive treatment with the Eluvia stent (n=309) or Zilver PTX stent (n=156). The primary efficacy endpoint was primary patency, defined as a peak systolic velocity ratio ≤2.4 on Doppler ultrasound, without CD-TLR or bypass of the target lesion. Lesions up to 140 mm in length could be treated. The Long Lesion Substudy enrolled patients with lesions >140 mm and ≤190 mm in length.40
At 12 months, non-inferiority was declared, with a primary patency of 86.8% (n/N=231/266) in the Eluvia stent group and 81.5% (n/N=106/130) in the Zilver PTX stent group. These data were later corrected to 92.1% primary patency for the Eluvia stent and 81.8% for the Zilver PTX stent. Freedom from target lesion revascularization was 95.5% for the Eluvia stent group and 91.0% for the Zilver PTX stent group (target lesion revascularization n=13 each, p=0.067). No deaths were reported in either group by the first year.40
The 2-year results were presented at LINC 2020, where freedom from target lesion revascularization was 86.5% in the Eluvia stent group (target lesion revascularization n/N=37/275, 13.5%) versus 79.9% in the Zilver PTX stent group (CD-TLR n/N=27/134, 20.1%, p=0.0495).37
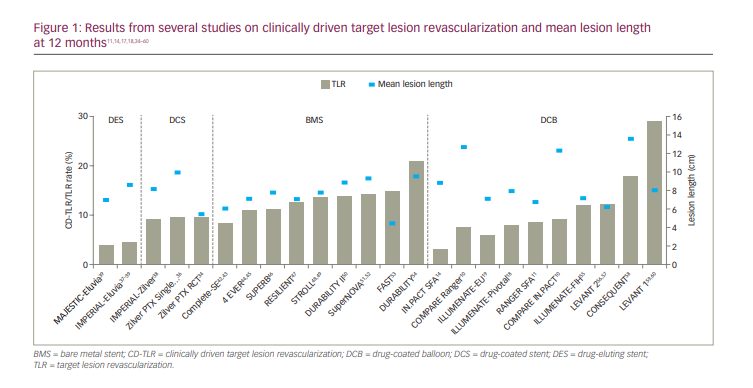
For an overview on other relevant trials, please refer to Figure 1.11,14,17,18,34–60
Other indications for use
Arteriovenous dialysis fistulae
Treatment of arteriovenous dialysis fistulae is challenging, as it commonly requires frequent interventions due to restenosis. Drug-eluting technologies help improve long-term results; nevertheless, it is worth noting that patients receiving dialysis tend to undergo multiple sessions of angioplasty of the arteriovenous fistulae, thus also potentially exposing these patients to larger doses of the drug eluted by the device at other organs, such as lungs.61
The LUTONIX AV (Study Comparing Lutonix AV Drug Coated Balloon vs Standard Balloon for Treatment of Dysfunctional AV Fistulae; ClinicalTrials.gov identifier: NCT02440022) was a randomized controlled trial in which 285 patients with arteriovenous fistula were randomized to receive either the LUTONIX® 035 DCB (Becton Dickinson, Maple Grove, MN, USA) or POBA.62 Target lesion primary patency at 180 days was 71.4% in the DCB group and 63.0% in the POBA group (p=0.0562). Statistical significance was not achieved, nevertheless, the overall results demonstrated a potential trend towards higher patency rates in the DCB group through 12 months. All-cause mortality at 12 months was 12.8% for the DCB group and 9.7% for the POBA group.63
Below the knee
Patients with critical limb ischaemia (CLI) typically have comorbidities, and are at increased risk of amputation and death compared with the normal population. Mortality is 20.0% at 6 months after diagnosis, and 50.0% at 5 years.64
The MERLION trial (Investigating the Safety and Efficacy of the Treatment With Luminor DCB and Angiolite DES of iVascular in TASC C and D Tibial Occlusive Disease in Patients With Critical Limb Ischemia; ClinicalTrials.gov identifier: NCT04073121) compared the Luminor DCB and Angiolite DES (both iVascular, Barcelona, Spain) in below-the-knee treatment of 50 patients with critical limb ischaemia (66 lesions).65 Data presented at LINC 2021 showed a freedom from target lesion revascularization of 82.0%, an amputation-free survival of 74.0% and primary patency of 69.0% after the first year. No deaths within 30 days of the procedure were reported.66
In the AcoArt II-BTK trial (ClinicalTrials.gov identifier: NCT02137577), 120 patients with CLI and infrapopliteal lesions received the Litos & Tulip® DCB (Acotec Scientific, Beijing, China) or the uncoated balloon (n=61 in the DCB group, n=59 in the POBA group).67 Freedom from CD-TLR at 12 months was seen in 91.5% of patients in the DCB group and 76.8% in the control group. No statistically significant difference in mortality (1.7% in DCB group versus 3.6% in control group; p=0.53) was detected up to 1 year.68
Discussion
Drug-eluting technology is a superior therapy for the treatment of peripheral arterial disease, when compared with uncoated balloon angioplasty. It has decreased the need for re-interventions by ensuring higher patency rates on a longer term. This has arguably helped to reduce costs (both for patients and hospitals, depending on location) and improve quality of life in patients with peripheral arterial disease.
Since the publication of a meta-analysis by Katsanos et al. in 2018 that showed higher pooled mortality rates in groups receiving paclitaxel-coated devices, healthcare agencies and societies have reviewed their guidelines on using such devices.5–8 As such, use of these devices has dramatically reduced, prompting research into drug-free devices.
To date, a definite dose–effect correlation between paclitaxel and mortality has not been established. The mortality signal has been inconsistent across several studies.
Recent follow-up data for long-term trials, such as the IN.PACT and ILLUMENATE randomized control trials, have also shown similar mortality rates between the DCB and POBA groups.14,15,17–19 An analysis of over 9 million claims from a German health insurance provider (64,771 patients, 107,112 procedures with 23,137 drug-eluting devices) could not find any association between increased mortality and the usage of paclitaxel-coated devices. Interestingly, DCB was associated with decreased mortality in the year following the procedure, with no notable difference in the years thereafter.69
New trials are focusing on substituting paclitaxel with alternative drugs, such as sirolimus.22 Nevertheless, the lack of consistent, positive data on these sirolimus-coated devices in previous studies still limits their application.
Currently, regulatory agencies and societies recommend that, until the increased mortality of patients who received paclitaxel-coated devices can be clarified, their use in clinical practice should be carefully evaluated and explicit patient consent should be obtained.7,8
It is the opinion of the authors of this article that the lack of both a drug dose–mortality relation and a causal explanation for the mortality risk defines an area requiring further investigation. Future endovascular device trial designs should incorporate strategies and methods to maximize patient retention and facilitate long-term assessment and reporting of vital statistics.
Paclitaxel-coated devices provide important benefits when used for endovascular therapy, especially in terms of restenosis and revascularization prevention, but may possibly increase fatality. To date, we continue to believe that the benefits outweigh the risks for use of these devices in selected patients who are at high risk for restenosis as determined by their treating physicians.