Over the past years, several reports have drawn attention to the need to consider sex-specific differences in cardiac resynchronisation therapy (CRT). Most importantly, there is growing concern on the underrepresentation of women in CRT landmark trials which laid the foundation for present guidelines. Patients with symptomatic heart failure, reduced left ventricular ejection fraction (LVEF) ≤35% and electrical conduction delay on the electrocardiogram (ECG) may benefit from CRT. International guidelines use a QRS duration entry criterion of 120 ms (USA) or 130 ms (Europe) before considering CRT.1,2 The level of recommendation depends on QRS morphology and QRS duration with a higher class of recommendation (i.e., class I indication) for patients with strict left bundle branch block (LBBB) morphology and QRS duration ≥150 ms. Although these cut-off values were derived from landmark trials with predominantly male subjects, the same criteria are currently used for women. Following these criteria, women are less likely to receive CRT, yet accumulating evidence suggests that they derive more benefit than men. Previous review-reports advocated for the existence of this sex paradox and called for new sex-specific guidelines.3–6 However, an overview of the mechanisms which explain sex differences in CRT outcome is currently lacking. This review summarises existing literature elucidating why sex-specific differences in CRT may exist.
The sex paradox
Females account for 17–33% of the study population in landmark trials.7–12 Half of these trials (MADIT-CRT, MIRACLE, RAFT) suggested greater effect of CRT on outcome in women. Others (CARE-HF, COMPANION, REVERSE) did not reveal a sex-related difference, although they may have lacked power to do this (Table 1). Two meta-analyses combined data from multiple landmark trials. Zusterzeel et al. pooled data from MADIT-CRT, RAFT, REVERSE comparing effects of a CRT defibrillator (CRT-D) versus an implantable cardioverter defibrillator (ICD) amongst 4,076 patients (22% women).13 In this study, women showed an overall favourable effect of CRT-D, and women with LBBB benefitted from CRT-D at a shorter QRS duration than men. Linde et al. pooled data from CARE-HF, MIRACLE, RAFT and REVERSE comparing CRT versus no CRT amongst 3,776 patients (21% women).14 Women tended to obtain greater benefit from CRT, but sex was not an independent predictor of clinical outcome.
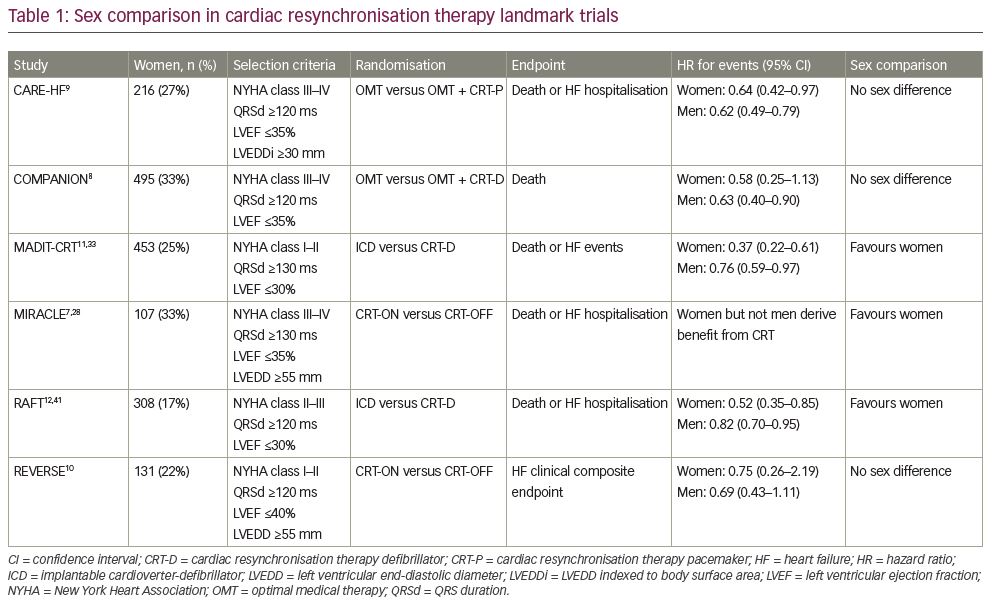
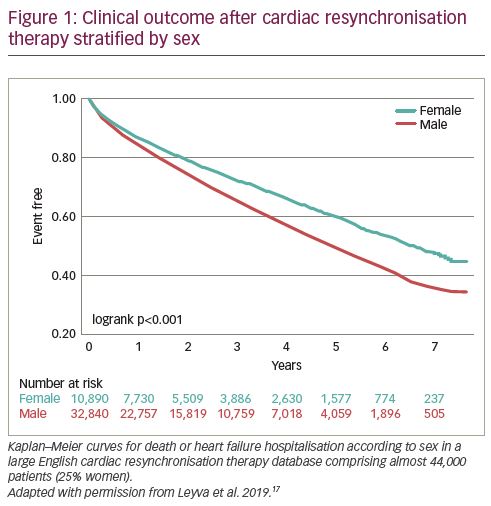
Over two decades, numerous studies have compared CRT outcome between women and men. Six meta-analyses and registries,15–20 each containing over 10,000 patients, consistently showed better clinical outcome for women, both on mortality and heart failure endpoints (Table 2). Leyva et al. recently published results from a large English database comprising almost 44,000 patients (25% women) confirming sex-specific differences in CRT outcome (Figure 1).17 Meanwhile, these data also underline the underrepresentation of women in the real-world CRT population. The Swedish Heart Failure Registry showed that female sex was an independent predictor of CRT underutilisation.21 Moreover, Chatterjee et al. showed, in a large cross-sectional study with over 300,000 patients, that sex differences in CRT-D implantation increased over time.22 These results have led to the general consensus that (i) women are less likely to receive CRT than men;
(ii) underutilisation of CRT in women results from using male-oriented guideline criteria (and a lower referral rate/higher refusal rate); (iii) women potentially derive more benefit from CRT than men.23
Natural history of heart failure
It should be considered that survival in patients with heart failure who receive CRT is dependent on (i) patient profile, and (ii) the effect of CRT. As most of the evidence comes from observational (i.e., non-randomised) studies, favourable outcome of women could be partially ascribed to a better natural history in heart failure (irrespective of the effect of CRT). In a large meta-analysis including almost 42,000 patients with heart failure without CRT (33% women), risk factors differed between men and women, with the latter being older and more likely to have hypertension but less likely to have a history of ischaemic heart disease or reduced LVEF.24 The main finding of this study was that survival is better for women with heart failure (hazard ratio [HR] 0.81, 95% confidence interval [CI] 0.78–0.85), irrespective of LVEF. Linde et al. performed a risk assessment for QRS width and morphology in nearly 15,000 patients with heart failure without CRT (28% women) and found that LBBB appeared to be harmful in men only.25 These results suggest that female CRT candidates may have a priori better survival, irrespective of CRT.
Heart failure aetiology
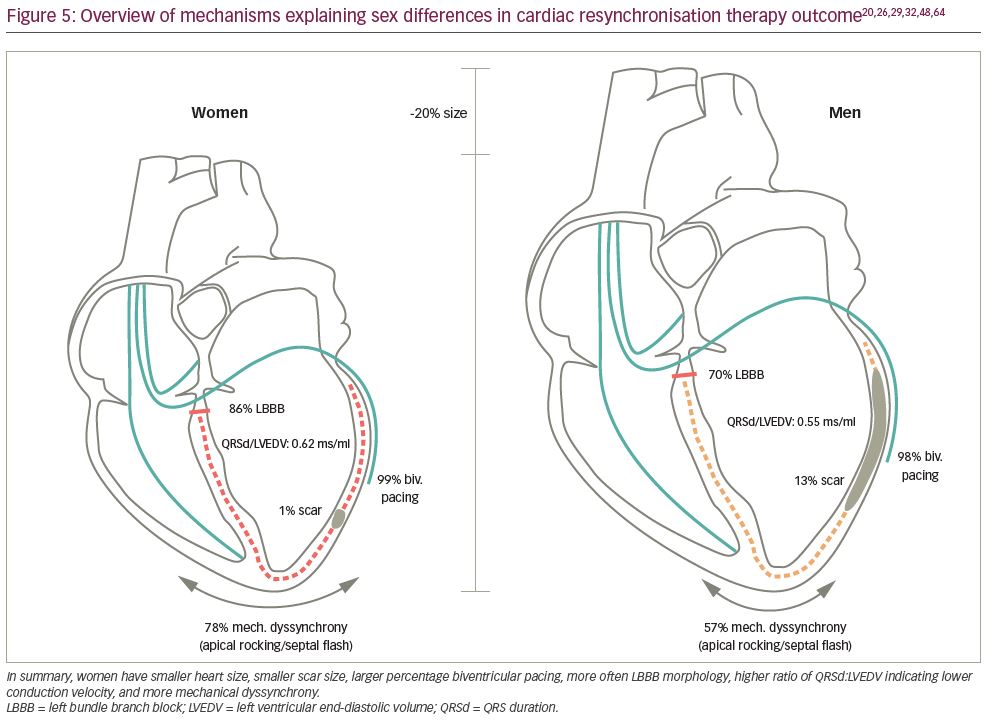
Ischaemic cardiomyopathy (ICM) is more prevalent in men, whereas women are more likely to have non-ischaemic cardiomyopathy (NICM). Loring et al. studied 235 patients (24% women) who underwent cardiac magnetic resonance (CMR) imaging before ICD or CRT-D implantation.26 Among the CRT subgroup, women had smaller myocardial scar size (1% versus 13%) and less transmural extents compared to men, particularly involving the inferolateral region (i.e., the target area for the left ventricular [LV] lead). In other words, women have more viable myocardium that can be recruited by CRT and less (ineffective) stimulation within scar tissue. Accordingly, patients with NICM have shown to exhibit greater reverse remodelling and better outcomes compared to those with ICM.27,28 Moreover, some studies suggested sex differences in CRT outcome to be almost entirely explained by differences in aetiology.29,30 In contrast, Leyva et al. showed in their large observational study of 44,000 patients (25% women) that sex-specific differences in CRT outcome persisted after separating groups by history of myocardial infarction.17 In the group of patients without myocardial infarction, females had significantly better clinical outcome compared to men, although no sex-specific differences were observed in the group with myocardial infarction. These results are in line with a study of Mooyaart et al. showing that within subgroups divided by sex and aetiology, women with NICM had the best survival rate.31 These findings suggest that differences in aetiology play a large, but not decisive, role in sex-specific differences in CRT outcome.
Delivery of therapy
The effect of CRT depends, among many factors, on device programming, the percentage of effective biventricular pacing, and LV lead placement. Cheng et al. sought to explore potential mechanisms of why sex differences exist, using data from 846 patients (33% women) from the SMART-AV trial.32 It was demonstrated that women derived greater reverse remodelling with CRT than men. These differences were explained by enhanced response to atrioventricular optimisation and greater degrees of biventricular pacing in women. Other studies confirmed a higher percentage of biventricular pacing in women, probably due to a lower rate of atrial fibrillation compared to men.13,31,33–35 Auricchio et al. found in the European multicentre CRT Survey II, containing over 11,000 patients (24% women), that women were more frequently implanted with a basal LV lead position and had larger reduction in QRS duration during CRT compared to men.34 In contrast, the distribution of the LV lead in the anterior, lateral and posterior position was comparable between sexes.
Mode of death
The mode of death in patients with heart failure can be defined as arrhythmic, pump failure or non-cardiac. Rho et al. studied a large cohort of 8,000 patients with heart failure (20% women) who potentially fulfilled eligibility criteria for implantation of an ICD for primary prevention of sudden cardiac death.36 Significant differences in mode of death were observed between sexes, with a lower proportional risk for sudden death (42% versus 50%) and higher proportional risk for pump failure death (36% versus 29%) in women compared with men. Given this finding, women will experience proportionately more benefit from CRT, which has been proven to significantly decrease mortality from pump failure deaths.7–12 In contrast, men are more likely to benefit from ICD implantation.37,38 These differences may be partly explained by the higher rate of NICM in women. Results from the randomised controlled DANISH trial with over 1,000 patients with heart failure (28% women) and the majority (58%) being equipped with CRT showed that prophylactic ICD implantation in patients with heart failure with NICM did not improve survival compared to standard care.39 These findings raise the question whether adding prophylactic ICD therapy to CRT implantation (i.e., CRT-D) provides any benefit over CRT only (i.e., CRT pacemaker) in women who pre-dominantly have NICM.17,35,40,41
Electrical dyssynchrony
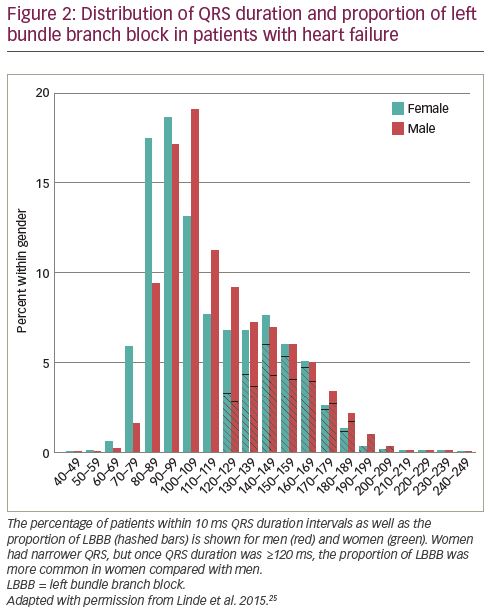
Normal QRS duration values are dependent on sex and race.42 Randolph et al. aimed to study QRS duration and morphology characteristics within the specific heart failure population amongst nearly 2,500 patients with reduced LVEF ≤35%.43 Although median QRS duration was ~5 ms shorter for women compared to men, LBBB was more common in women (24% versus 14%). These results indicate that at any given QRS duration, women might exhibit more substrate for electrical resynchronisation than men. This was confirmed by Linde et al. showing that women had LBBB morphology at narrower QRS duration (Figure 2).25 Using identical QRS duration cut-off criteria for men and women will therefore lead to the selection of women with more severe conduction disorder (i.e., true LBBB). In line with this concept, three large echocardiographic CRT studies (each containing over 1,000 patients) demonstrated that women more frequently have mechanical dyssynchrony (septal flash or apical rocking) at shorter QRS duration compared to males.29,44,45 This is an important finding as the correction of inefficient mechanical contraction underlies LV pump function improvement after CRT.
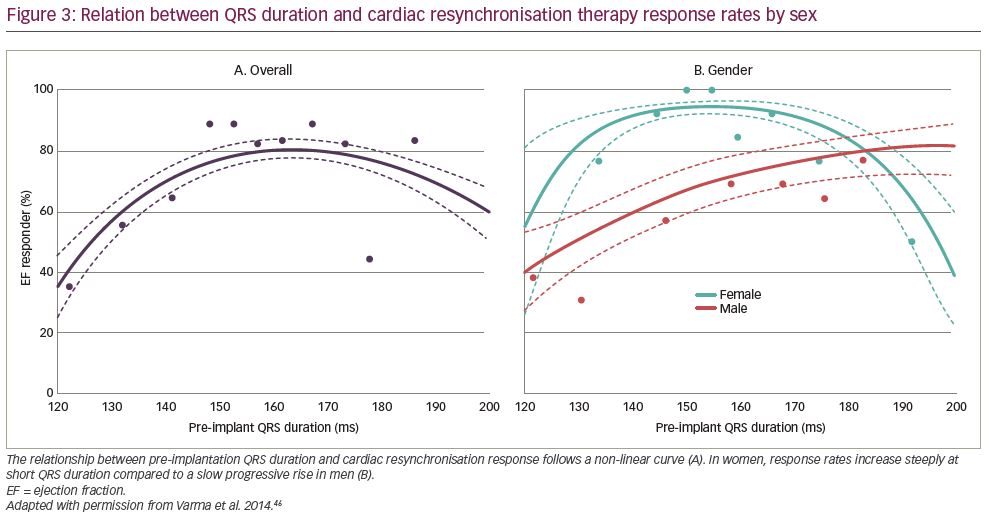
Varma et al. compared QRS duration–CRT response relations in 212 patients (50% women) with NICM and LBBB.46 Women showed a different QRS duration–CRT response relation than men with relatively more benefit at shorter QRS duration (particularly <150 ms) (Figure 3). Moreover, multiple studies evaluated the prognostic significance of LBBB detection according to sex. The largest of these studies was performed by Loring et al. by analysing Medicare billing data of almost 145,000 CRT patients (26% women) in the USA.18 LBBB diagnosis was associated with a 26% mortality reduction in women (HR 0.74, 95% CI 0.71–0.77) and a 15% mortality reduction in men (HR 0.85, 95% CI 0.83–0.87) with a significant interaction between sex and LBBB. In addition, Zusterzeel et al. studied nearly 32,000 CRT-D recipients (36% women) from the USA national cardiovascular data registry and found that women had a 21% lower mortality risk than men (HR 0.79, 95% CI 0.74–0.84); however, there was no difference in non-LBBB patients (HR 0.95, 95% CI 0.85–1.06).20 Lastly, women with LBBB in the MADIT-CRT study achieved a significantly better result with CRT than their male counterparts.33,47 In summary, considerable differences are observed in women regarding the manifestation of substrate for electrical resynchronisation (at shorter QRS duration) and the prognostic significance of LBBB diagnosis (more benefit from CRT). These differences may be explained by differences in heart size as will be discussed in the following section.
Heart size
In healthy subjects, women demonstrate up to 20% smaller LV dimensions compared to men.48 Throughout the heart failure course, women generally maintain relatively normal LV size whereas men suffer from adverse cardiac remodelling, further amplifying differences in heart size.49 Theoretically, QRS duration is determined by both myocardial conduction velocity (i.e., substrate) and travel distance of the wave front (i.e., heart size). In line with this concept, a positive correlation exists between LV volumes or mass and QRS duration in both narrow QRS and LBBB.50 De Pooter et al. demonstrated that LV size was an independent predictor of LBBB-QRS duration among more than 1,000 CRT patients (41% women).44 Moreover, Lee et al. recently showed with computer simulations that LV size accounts for 50–100% of the QRS duration differences between CRT candidates.51 Sex differences in heart size will thus lead to considerable longer QRS duration in men.52
Although QRS duration prolongation is expected to result in improved CRT benefit, advanced LV dilation is actually associated with poor CRT outcome.53–56 Guo et al. showed that a wide QRS complex better predicted CRT response in absence of LV dilatation;57 however, these results were contradicted by Rickard et al.58 Linde et al. found in a meta-analysis of five randomised clinical trials, that accounting for patient height improved predictive value of QRS duration for clinical outcome after CRT, although part of this effect may also be ascribed to differences in heart size.14 As heart size can be considered a confounder in the assessment of electrical dyssynchrony between men and women, normalisation of QRS duration to LV size may be considered to improve patient selection for CRT.
Several studies have called for new sex-specific CRT guidelines, either by lowering QRS duration cut-off values by ~10 ms for women,3–6,44,51,59 or by providing a higher class of recommendation (i.e., class I indication) in women with LBBB and QRS duration <150 ms.13,20,46,47 Although these measures are likely to improve patient selection for women, men would still be subject to overtreatment. Moreover, simply subtracting a fixed 10 ms in women may not be sufficient to equalize sex-differences as this ‘frameshift’ would not superimpose the QRS duration–CRT response curves as shown in Figure 3.46
Recently, we proposed an alternative method to improve patient selection in both men and women by showing that normalising QRS duration to LV dimension, to adjust for differences in heart size, improved prediction of CRT response.60 In this proof-of-concept study, 32 patients (38% women) underwent pre-implantation CMR imaging followed by invasive pressure–volume loop measurements directly after CRT implantation. It was demonstrated that unadjusted QRS duration was unrelated to haemodynamic changes during CRT, whereas normalisation of QRS duration to LV dimension resulted in a good correlation with acute haemodynamic CRT response. Women displayed a higher QRS duration:LVEDV ratio (due to smaller ventricles) indicating slower conduction at baseline, and showed more LV pump function improvement during CRT compared to men. In a subsequent study by Varma et al. with 130 patients (55% women), sex-specific differences in the QRS duration–response relationship (Figure 3) resolved after QRS duration was normalised to LV dimension.61 In addition, analysis of 787 patients (28% women) with narrow QRS duration (<130 ms) in the ECHO-CRT study showed that most of the harmful effects of CRT were observed in men with a low QRS duration:LVEDV ratio, whereas women with a higher QRS duration:LVEDV ratio exhibited significant reverse remodelling.62,63
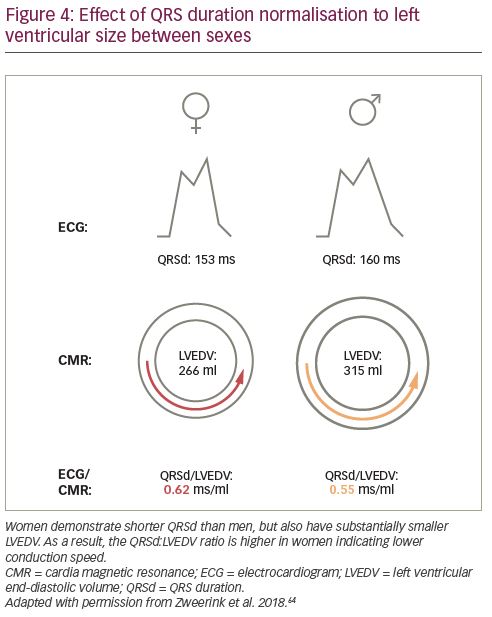
Most recently, the role of QRS duration normalisation was extended to the prediction of clinical outcome in a large two-centre study including 250 patients (34% women) who underwent pre-implantation CMR imaging.64 First, normalisation of QRS duration to LV dimension improved prediction of survival after CRT implantation in the total population. Second, women demonstrated shorter QRS duration than men but also substantially smaller heart size, resulting in a higher QRS duration:LVEDV ratio as illustrated in Figure 4. Third, sex-specific differences in CRT outcome were found to be explained by normalised QRS duration, rather than sex itself, in multivariable analysis.
Conclusions
Multiple mechanisms can be held accountable for the sex difference in CRT outcome as summarised in Figure 5 and Table 3. Women exhibit different risk factors resulting in better heart failure prognosis, irrespective of CRT. They more often display factors associated with good response to CRT such as NICM with lower scar burden and LBBB at shorter QRS duration, possibly due to smaller heart size. Conflicting evidence exists as to whether differences in CRT outcome can be attributed to these factors,14,16,29,30,33,54,57,61,64 or if after correction a true sex difference exists.17,18,31,35,41,45,47,58 Possibly, other biological factors such as hormonal differences and disparities in cardiac autonomic physiology come into play.65 Ultimately, understanding sex differences plays a pivotal role for the development of new strategies to equalize sex differences in CRT such as normalisation of QRS duration to LV size. Prospective studies are needed to evaluate the effect of these measures.