Percutaneous coronary intervention (PCI) has evolved over recent years, with increasingly more complex, high-risk procedures being performed, including in patients with multivessel coronary artery disease with or without left main disease, often complicated by severe left ventricular (LV) dysfunction.1 In addition, decisions about coronary bypass surgery or PCI often focus not only on coronary anatomy, but also on clinical factors including renal impairment, age and body mass index (BMI), as well as patient wishes. For many patients considered poor candidates for coronary bypass surgery due to significant comorbidities, PCI represents an appropriate alternative.
A recent analysis found that 21.5% of potential patients for coronary bypass surgery patients were deemed ineligible for surgery and underwent PCI.2 PCI for such patients (often with impaired LV function) involves an elevated risk for circulatory collapse during the procedure. Such procedures are considered high-risk PCI (HR-PCI), where protective mechanical circulatory support (MCS) may be beneficial during the PCI procedure.3,4 The intra-aortic balloon pump and the percutaneous LV assist device (pVAD) are the two most frequently used MCS devices for HR-PCI. The intra-aortic balloon pump chiefly serves to increase arterial pressures through counterpulsation, with limited improvement in cardiac output. In contrast, micro-axial rotary pumps, or pVADs, remove blood from the left ventricle and pump it into the ascending aorta, decreasing LV filling pressures and providing forward flow cardiac output.5 The Impella® 2.5 and CP heart pumps (Abiomed, Danvers, MA, USA) are the most commonly used pVADs for HR-PCI, with increasing use over the last decade.6 Though the randomised PROTECT II trial showed equivalent outcomes with Impella and intra-aortic balloon pump-supported HR-PCI at 30 days (the primary endpoint),7 there was a trend toward a late reduction in major adverse events for the Impella support group at 90 days, which attained significance when the analysis was conducted on the per-protocol population.8 Postulated benefits of pVAD haemodynamic support are to provide operators time to perform more complete revascularisation.9
Despite the potential benefits of pVAD support, bleeding and vascular complications remain a concern for elective use in HR-PCI, largely related to large-bore vascular access. Such pVAD-related vascular complications are linked with significantly higher in-hospital mortality, length of stay and cost,10 limiting the use of prophylactic pVAD circulatory support. However, the observed rate of bleeding complications with Impella use has shown significant variability in published studies, which are largely retrospective and/or observational in nature. A recent analysis by Amin et al. including cardiogenic shock, a markedly higher-risk population compared with HR-PCI, identified 2.5 times variability for Impella bleeding complications across centers.6 Thus, the true risk of bleeding and vascular complications with Impella use remains incompletely defined.
Major bleeding and vascular complications with Impella-supported high-risk percutaneous coronary intervention
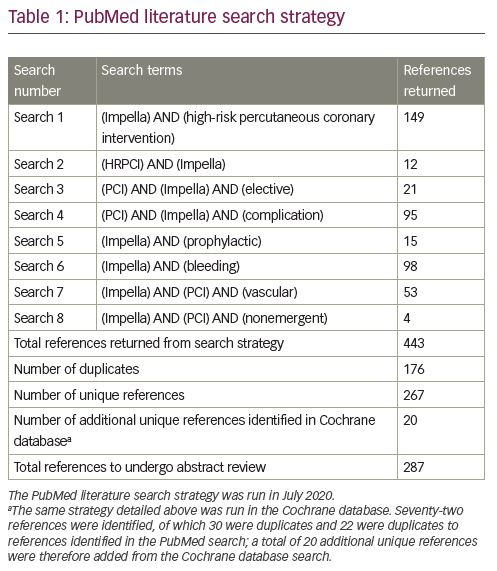
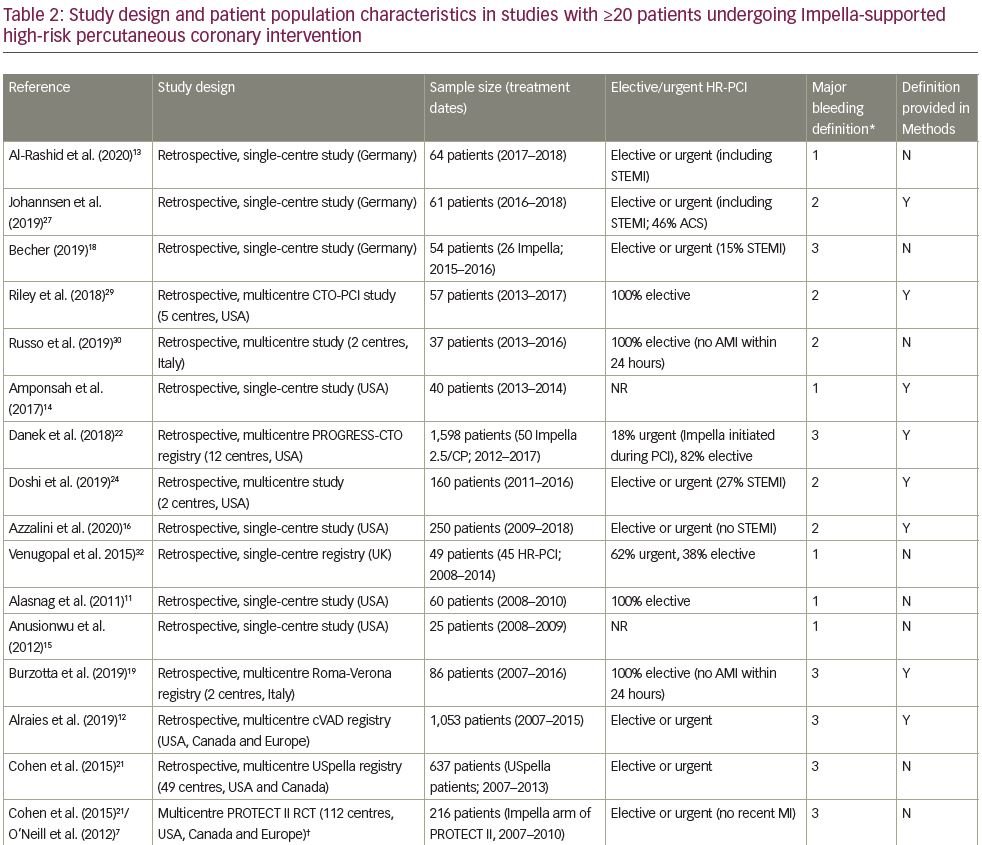
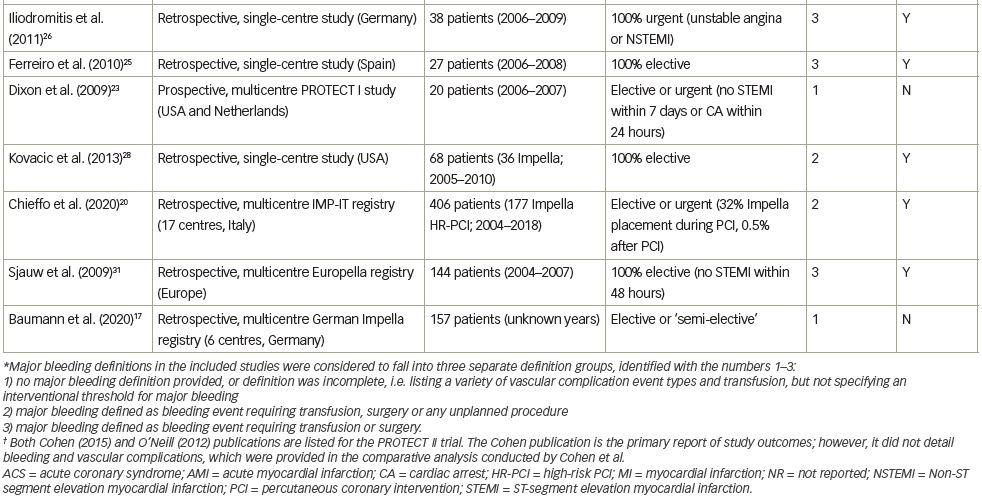
To evaluate the risk of major bleeding and vascular complications in Impella-supported HR-PCI, we performed a comprehensive literature review. The literature search was conducted in the PubMed and Cochrane databases in July 2020. The full literature search strategy is available in Table 1. Our review was limited to studies reporting results of ≥20 patients,7,11–32 in an attempt to capture studies from centres with more than casual experience with large-bore access and Impella. We identified 23 such studies involving 3,466 patients undergoing elective or urgent HR-PCI, with Impella support initiated before or during PCI (Table 2).7,11–32 The dataset comprises two prospective trials (the PROTECT I feasibility trial and the randomised PROTECT II pivotal trial); 9 retrospective, multicentre studies/registries; and 11 retrospective, single-centre studies. Major bleeding complications and major vascular complications were recorded per study report, though the reports were subject to varying definitions of major bleeding or vascular complications. Nearly half of the studies (10 studies) did not directly specify a bleeding complication definition. Such variability of definitions precluded a formal meta-analysis; thus, our review is qualitative, describing the published experience of bleeding and vascular complications consistent with
real-world experience.
Major bleeding
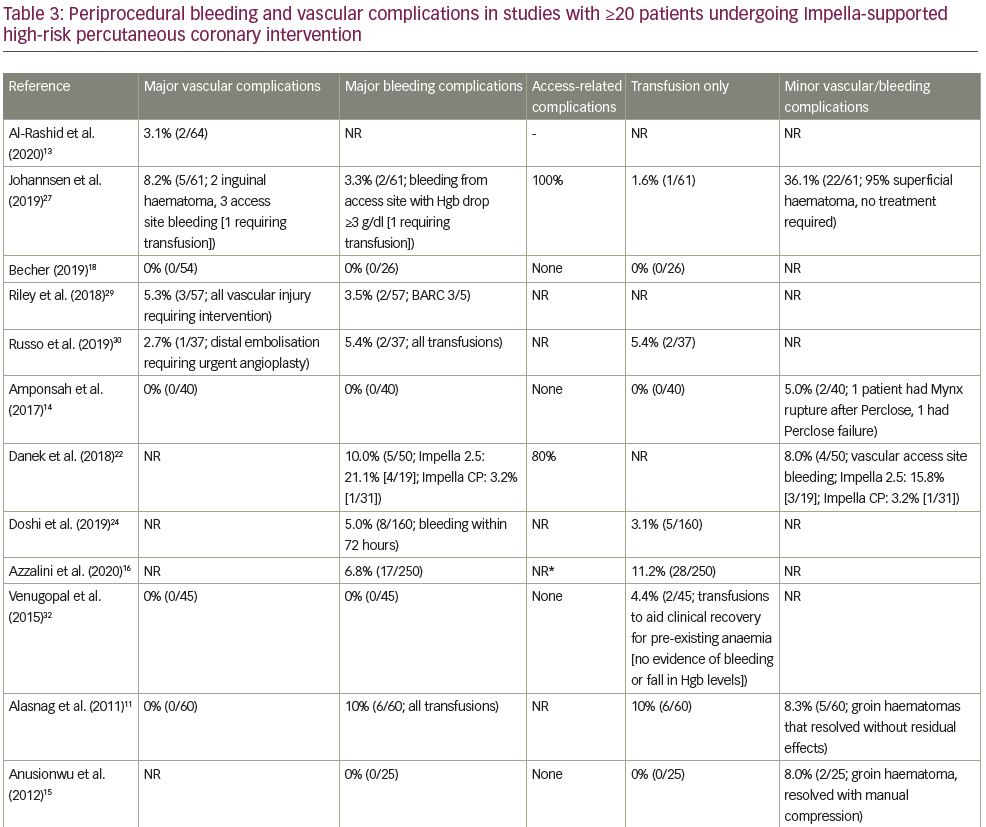
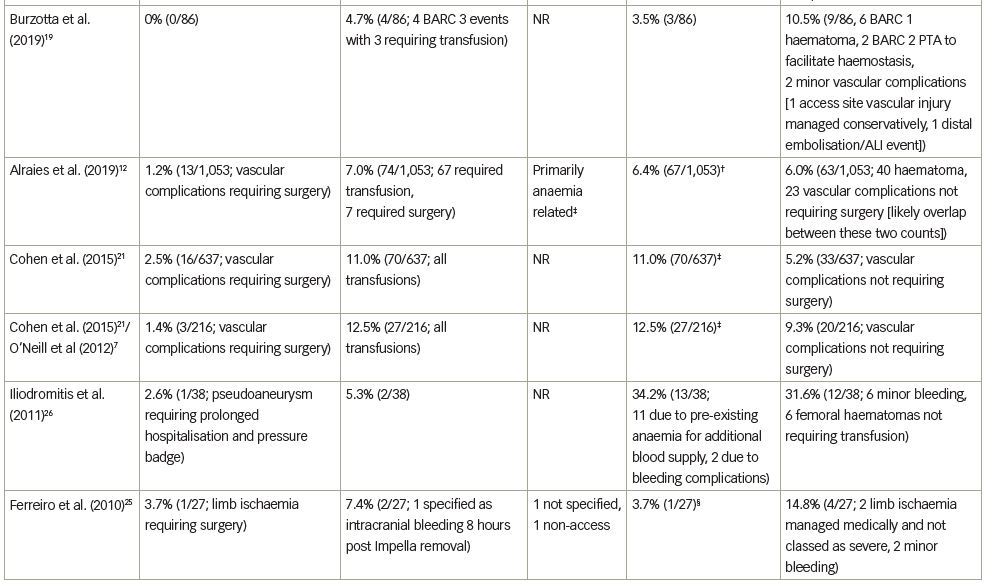
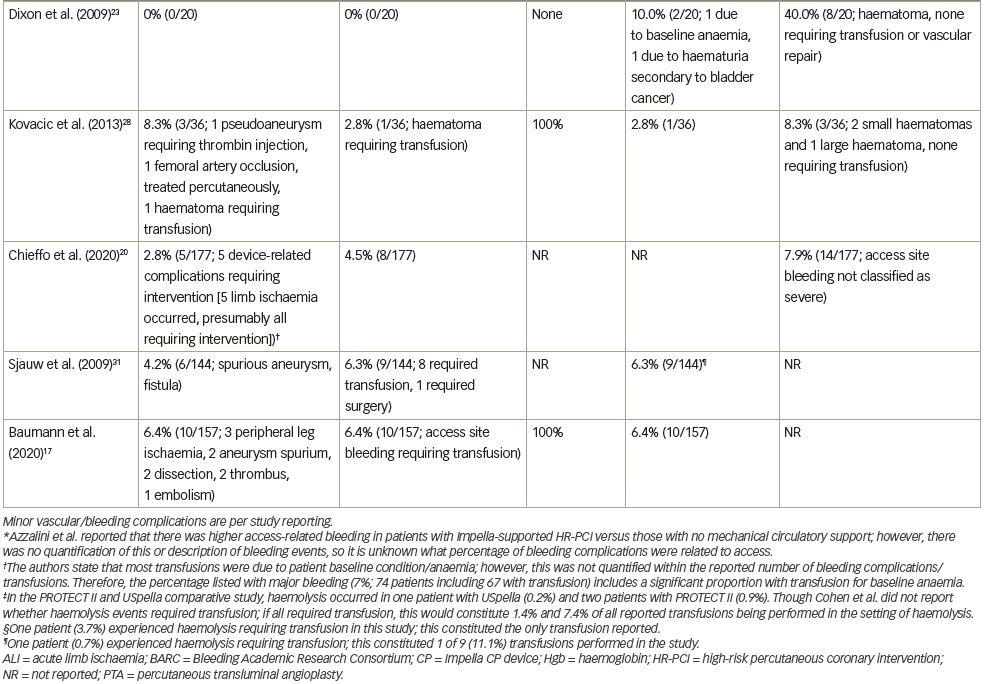
The median rate of major bleeding complications was 5.2% (range, 0–12.5%), with 22 of 23 studies contributing data. The rate of bleeding complications varied widely, likely due to variable definitions, incomplete data in some studies relative to site-specific bleeding, inclusion of non-groin-related bleeding events and occasionally the inclusion of emergent, ST-segment elevation myocardial infarction (STEMI) cases. Twelve studies (54.5%) reported major bleeding events without specifying the bleeding site.7,11,16,19–21,24–26,29–31 Five studies (22.7%) reported that no major bleeding complications occurred.14,15,18,23,32 The remaining five studies (22.7%) reported on bleeding complications and specified the bleeding site.12,17,22,27,28 Of the five studies reporting bleeding complications and bleeding site, three studies reported that 100% of major bleeding complications were access-related, with major bleeding complication rates of 2.7%, 3.3% and 6.4% reported in these three studies.17,27,28 One study reported that 80% of major bleeding complications were access-related.22 In the other study (a retrospective, gender-based analysis of 1,053 patients treated in the multicentre cVAD registry), major bleeding complications occurred in 7.0% of patients, with 90.5% of bleeding events consisting of blood loss requiring transfusion.12 The authors noted that low baseline haemoglobin levels and anaemia were mainly responsible for the elevated bleeding complication rate observed in females, as vascular complication rate was low (1.2%) and similar between genders.
Transfusion rates
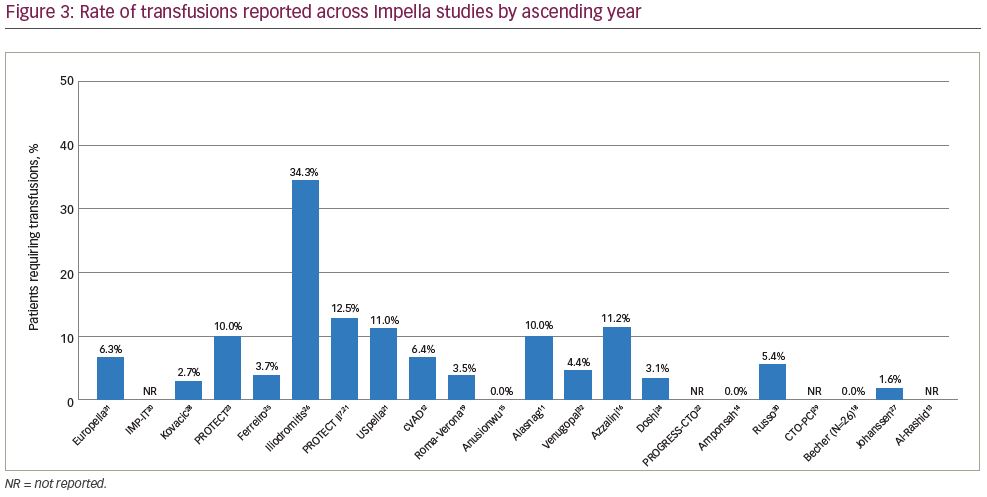
Transfusion rates showed the largest variation across studies, with a median rate of 5.4% (range, 0–34.3%) and 19 studies contributing data. In some studies, the number of patients requiring transfusion exceeded the number of reported bleeding complications, which likely reflected transfusions for baseline anaemia in some cases. Transfusion rates in the four studies in which the majority of transfusions were stated as being performed in the setting of baseline anaemia ranged from 5.0% to 34.3%.12,23,26,32 Few studies included in our review reported on haemolysis (9 of 23 studies). Of these, 3 studies reported 0% haemolysis rates, 1 reported only mild haemolysis not requiring treatment, and 5 reported haemolysis occurring in one or two patients; overall haemolysis rates in studies reporting any hemolysis events ranged from 0.2% to 3.7%. Haemolysis requiring transfusion did not contribute meaningfully to overall transfusion rates in most cases (Table 3).
Vascular complications
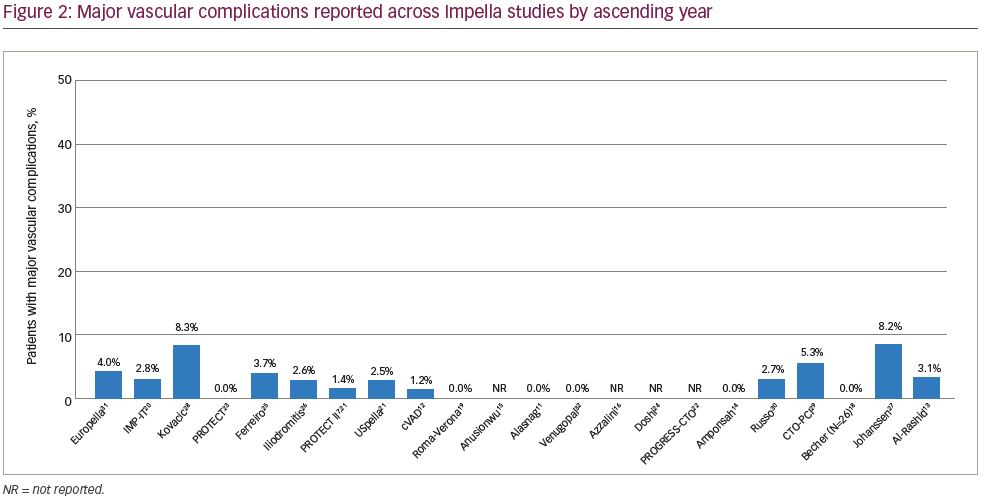
Median rate of major vascular complications was 2.6% (range, 0–8.3%), with 15 of the 19 studies reporting a vascular complication rate <5%. The rate of vascular complications did not vary as widely as bleeding complication rates; this finding likely reflects the fact that major bleeding complications may stem from non-access as well as access-related causes, whereas vascular complications, by definition, are more specific.
Trends in bleeding and vascular complications over time
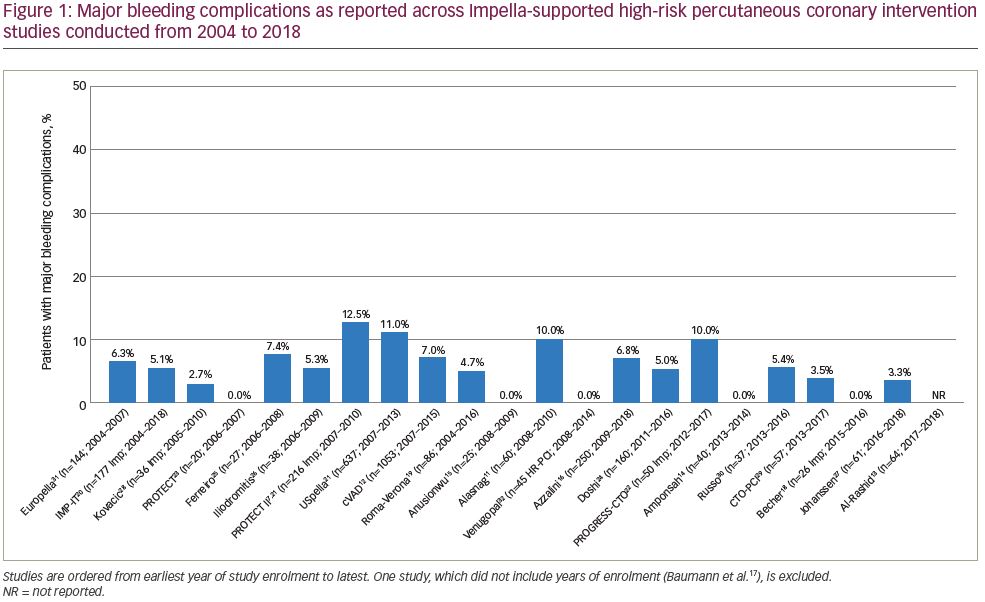
Bleeding complications over time demonstrated that the variability in bleeding risk seen across centres has not changed meaningfully over the last 15 years (Figure 1).7,11–16,18–32 Given the broad scope of bleeding complications (comprising both access and non-access–related bleeding during PCI), often complicated by pre-existing anaemia and the inclusion of some urgent HR-PCI patient populations, this variability is not surprising. Likewise, many series reported relatively low numbers of cases over extended time periods, suggesting that any advantages of increased operator skill, using best practices accrued through increasing experience, could not be ascertained. While we attempted to focus on higher-volume reports (≥20 patients) to provide ‘best practice’ results, eliminating ‘occasional users’, even in larger studies it is impossible to identify the extent to which specific operators or teams were involved in a series of cases, minimising our ability to assess whether any potential learning curve with the large-bore Impella procedure is driving bleeding complication rates. Thus, low yearly volumes may obscure the ability to ascertain whether improvements in outcomes over time occur with increased operator and institutional experience for large-bore access and supported HR-PCI, as has been reported for trans-aortic valve repair (TAVR).33,34 The rate of vascular complications remained consistently low over time (Figure 2).7,11–16,18–32 Transfusion rates suggest a downward trend over time (Figure 3).7,11–16,18–32
Impact of access and non-access bleeding complications on clinical outcomes
A significant limitation in published studies on HR-PCI is the relative rarity of studies identifying the specific aetiology of bleeding complications – whether access-related or gastrointestinal bleeding, etc. In a large CathPCI registry analysis of bleeding in a largely elective-urgent, not specifically high-risk PCI patient population, Rao et al. reported a similar overall bleeding rate of 5.8% and found that 32% of bleeding events had a specified site, with a further 44.6% due to haemoglobin decrease and 21.8% due to blood transfusions.35
The prognostic relationship of PCI bleeding complications with mortality varies greatly depending on the site of bleeding. A large meta-analysis of PCI studies (primarily acute myocardial infarction patient populations) specifying the site of bleeding complications found that, while both access and non-access–related bleeding complications were linked with an increased risk for mortality, the risk ratio (RR) for non-access site bleeding was considerably higher than with access site bleeding (RR 4.06 and 1.71, respectively).36 To our knowledge, there has been no comprehensive review of access versus non-access–related bleeding complications and their respective prognostic values in an exclusively elective-urgent HR-PCI population with MCS support. Our review identified the difficulties in conducting such a review from the current literature, as a majority of studies did not specify bleeding site.
The bleeding definition used also significantly impacts the prognostic value of reported bleeding events, and in our review, varied widely across published studies. Often, major bleeding definition was not specified at all. A meta-analysis of PCI studies (STEMI and elective patient populations) encompassing several different bleeding definitions found that, depending on the definition used, the mortality risk from a major bleeding event ranged from a 1.5 to 6.7 times increase.37
Factors impacting bleeding complications in Impella-supported high-risk percutaneous coronary intervention
An observed increase in femoral access complications with PCI in recent years may be due, in large part, to the swift uptake of radial access for conventional PCI procedures by many operators.38 This ‘Campeaux Paradox’ has occurred due to operators having fewer femoral access cases, with a subsequent loss of femoral skills. Though radial access for PCI has been associated with reduced complications compared with femoral access in several prospective studies,39 there will always be a requirement for femoral access in some cases with challenging anatomy, necessitating that operators maintain femoral skills, both for PCI access and for large-bore sheath procedures, such as TAVR and pVAD placement. There is also a potential learning curve for large-bore access procedures, not only on the operator level but also involving team experience with large-bore procedures and effective management of access complications. In the USpella registry, Cohen et al. identified declining transfusion rates of 12.2%, 7.4% and 6.1% in patients treated in the years 2009, 2010 and 2011, respectively.21 The authors suggested that a learning curve with large access closure techniques was potentially responsible for the decreasing transfusion rate over time.
In multiple studies identified in this review, all, or a majority of, transfusions were to aid clinical recovery due to baseline anemia.12,23,26,32 Low baseline haemoglobin levels were one of 10 variables included in a validated bleeding risk model developed by Rao et al. to delineate high bleeding risk in patients up to 72 hours after undergoing PCI.35 While some factors were related to clinical circumstances, such as acute myocardial infarction and/or shock, others were patient-related and likely risk factors related to Impella procedures including female gender, age >70 years, renal failure, diabetes and high BMI. Thus, bleeding/transfusion risk is a composite of access site and clinical patient features. Those patients deemed to be high bleeding risk merit careful consideration of whether MCS is necessary to support the HR-PCI procedure. Emerging technologies aimed to reduce vascular complications with large-bore access may ameliorate the risk of access-site bleeding and vascular complications in patients with high bleeding risk, and are discussed in a later section.
Risk–benefit assessment of elective Impella support
Considerations for MCS haemodynamic support vary by severity and complexity of anatomic coronary disease, overall patient clinical risk and baseline haemodynamic stability including overall LV function, further modulated by operator-estimated patient risk for haemodynamic collapse during PCI. While operator experience is often considered a factor, not all studies have shown an outcome benefit for high operator experience.40 Bleeding and vascular complications remain an important consideration related to MCS support for HR-PCI.
An important consideration for operators is the outcome risk when managing a patient with acute haemodynamic collapse during PCI, most often secondary to a complication. A recent cVAD registry analysis of patients undergoing HR-PCI with Impella, either pre-procedure (prophylactic) or as bailout, showed a striking risk for the bailout group.41 Specifically, those patients treated with bailout as opposed to prophylactic Impella support during HR-PCI had significantly higher in-hospital mortality (49.1% versus 4.3%).41 What remains unknown is the percentage of patients undergoing unsupported HR-PCI who ultimately experience severe haemodynamic instability requiring bailout MCS. Thus, in the absence of Level 1 evidence and specific guidelines for pVAD support during HR-PCI, variation in approaches further add to the difficulty in analysing overall procedural risk as well as bleeding risk.
Limitations of clinical dataset and future directions
In the studies we reviewed, the site of bleeding events was seldom specified. Importantly, reported bleeding complications not only reflect complications due to large-bore Impella access, but other access sites such as PCI access, in addition to gastrointestinal bleeding, transfusions to aid clinical recovery, anticoagulation practices (i.e. failure to titrate total heparin administration for the Impella purge solution), patients with high bleeding risk, or other, unspecified causes.
In recent years, there has been significant focus on developing improved techniques for access site and anticoagulation management, as well as appropriate closure techniques, all potentially limiting access-site bleeding risk. Few studies have been conducted exclusively in the last 5 years on Impella-supported HR-PCI with reported outcomes on complications observed. In fact, no studies of ≥20 patients appear to have been conducted exclusively in the last 2 years. This limits our ability to assess the impact of emerging solutions for large-bore access strategy on bleeding complication rates.
Evolving solutions
In an attempt to reduce bleeding risk, a variety of new techniques continue to be introduced. Because the number of vascular access sites required for a complex procedure increases the risk of a vascular event, a newly introduced single-access Impella technique has the potential to reduce access-site complications for Impella-supported HR-PCI by eliminating the need for a separate PCI access site.42 While an initial report on outcomes with this technique is encouraging, more data are needed to assess its safety and efficacy.42 Recent reports suggest that the use of routine ultrasound guidance for large-bore femoral access may reduce vascular and bleeding complications.43,44 Another development for large-bore procedures is the recent approval of the MANTA® VCD (Teleflex, Morrisville, NC, USA), a closure device specifically designed for 10–20 Fr large-bore procedures, though predominantly evaluated in TAVR studies published to date.45–48 While new techniques and devices are encouraging, larger studies will be required to both identify and confirm the optimal strategies to ultimately reduce bleeding and vascular complications associated with large-bore access for haemodynamic support in HR-PCI.
Conclusions
Bleeding and vascular complications adversely affect clinical outcomes after Impella-supported HR-PCI. Rates of major bleeding complications have shown wide variation over the last 15 years, underlining the importance of recognising the risks related to large-bore sheath procedures. Transfusion rates appear to have decreased over time, whereas occurrence of major vascular complications has remained relatively constant (<5% in most studies over the last 15 years). Thus, patient selection for protected HR-PCI should involve consideration of high bleeding risk factors and utilising currently accepted, patient-specific, bleeding avoidance strategies, as well as optimal vascular access management to minimise bleeding and vascular risk when Impella is used for HR-PCI.