Since enalapril was shown to reduce the risk of all-cause death in the CONSENSUS1and SOLVD-T trials,2angiotensin-converting enzyme inhibitors (ACEIs) have been the mainstay of treatment for patients with heart failure (HF) and a reduced ejection fraction (HFrEF). In the SOLVD-T trial there was a relative risk reduction (RRR) of 16% in all-cause death and of 26% in HF hospitalisation among patients with mild-to-moderate symptoms with long-term enalapril treatment.2Further studies showing additional reductions on mortality and HF hospitalisation by the subsequent addition of beta-blockers (BB) and mineralocorticoid receptor antagonists (MRA) to ACEIs, established triple neurohormonal blockade as the current paradigm of medical therapy in HFrEF.3-6However, there is an unmet need to further reduce mortality and morbidity risk; 5-year mortality risk among HF patients is >50%.7
The current therapeutic strategy in HFrEF is based on the antagonism of the regulatory systems (renin-angiotensin-aldosterone system and the sympathetic nervous system among others) that produce vasoconstriction, sodium and water retention, hypertrophy, apoptosis and fibrosis, all of them having a negative impact on ventricular remodelling and prognosis in the long term.8To improve further ventricular remodelling and clinical outcomes a complimentary strategy would be the stimulation of the counter-regulatory systems (natriuretic peptides [NPs] system and others) which have effects that oppose those of the regulatory systems. LCZ696 (Entresto™, Novartis AG, Basel, Switzerland) is a drug that antagonises the regulatory systems and, in addition, stimulates the counterregulatory systems.
LCZ696 comprises the neprilysin inhibitor, sacubitril (AHU377) and the angiotensin receptor blocker (ARB), valsartan.9,10Neprilysin is a neutral endopeptidase that degrades a range of endogenous vasoactive peptides,11,12among them NPs and angiotensin–II. Inhibition of neprilysin by sacubitril therefore increases the levels of NPs providing beneficial haemodynamic and molecular effects. However, sacubitril, when used alone, also increases the levels of angiotensin–II, countering the beneficial effects derived from NPs levels increase. Therefore, to obtain full benefit from sacubitril an ARB must be added to block angiotensin receptor-1 stimulation by the increased levels of angiotensin-II.13,14
The addition of counter-regulatory systems stimulation to the traditional strategy of regulatory systems blockage constitutes a new paradigm in HF treatment that was evaluated in the PARADIGM-HF study. The hypothesis was tested that LCZ696 would be superior to enalapril in reducing cardiovascular mortality and morbidity in patients with HFrEF. When considering the ethical aspects of not using an ACEI in the LCZ696 arm it is relevant to mention that the latter contains a valsartan moiety and that ARBs remain recommended as an alternative to ACEIs in patients with HFrEF.15
PARADIGM-HF
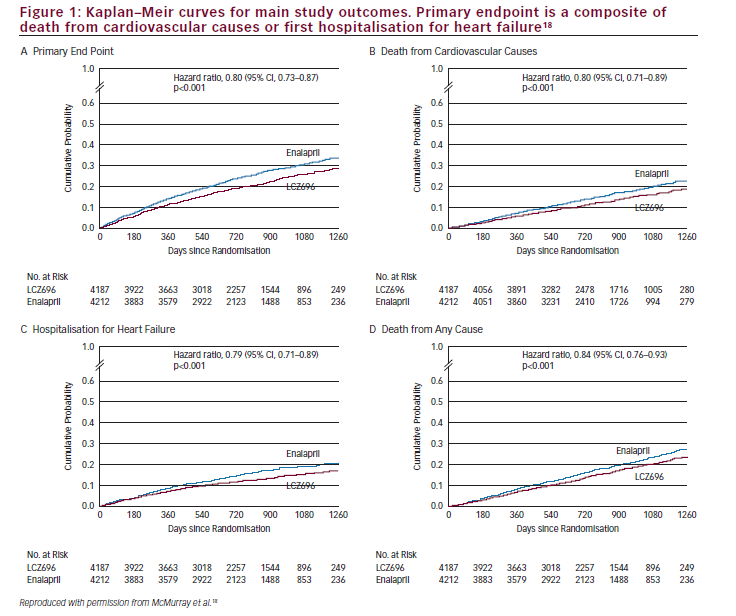
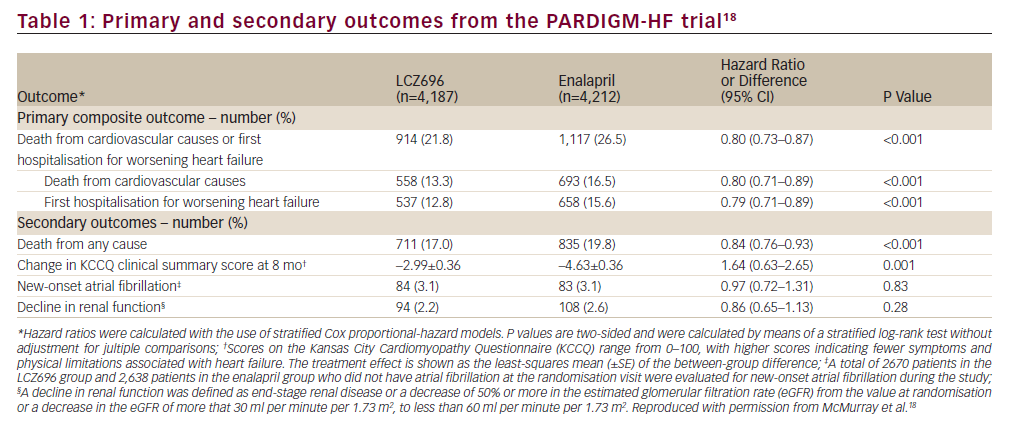
In the randomised, double-blind, multicentre, international trial PARADIGM-HF (ClinicalTrials.gov number NCT01035255), 8,442 patients with HF and a left ventricular ejection fraction ≤40%, in New York Heart Association (NYHA) class II–IV were randomised to either LCZ696 200 mg twice daily or enalapril 10 mg twice daily in addition to recommended therapy, including BBs and MRAs.16,17The boundary for an overwhelming benefit of LCZ696 was crossed which meant that, according to pre-specified rules, after a median follow up of 27 months, the trial was stopped early.18Primary outcome, which was a composite of death from cardiovascular causes or a first hospitalisation for HF, occurred in 914 patients (21.8%) in the LCZ696 group compared with 1117 patients (26.5%) in the enalapril group (95% confidence interval [CI], 0.73 to 0.87; p=0.0000002, number needed to treat [NNT]=21) (Figure 1A and Table 1).17,19
Both components of the primary composite endpoint were reduced by LCZ696 compared with enalapril: time to cardiovascular death (hazard ratio = 0.80 [95% CI: 0.71–0.89]; p=0.00004, NNT=32) (Figure 1B and Table 1) and time to first HF hospitalisation (hazard ratio = 0.79 [95% CI: 0.71–0.89]; p=0.00004, NNT=36) (Figure 1C and Table 1).17,19LCZ696 was more efficacious than enalapril both in terms of reducing sudden cardiac deaths and reducing deaths from worsening HF; this accounted for the majority of cardiovascular deaths.20The LCZ696 treatment effect for sudden cardiac death was not influenced by the presence of defibrillator devices.20 The efficacy of LCZ696 was superior to that of enalapril across the spectrum of age of the PARADIGM-HF patients, with a favourable benefit–risk profile apparent in all age groups.21Although most PARADIGM-HF patients had mild symptoms, many were at high risk for adverse outcomes and obtained a large absolute benefit from LCZ696, compared with enalapril.22 In addition, in surviving patients with HF, LCZ696 prevented clinical progression more effectively than enalapril.23
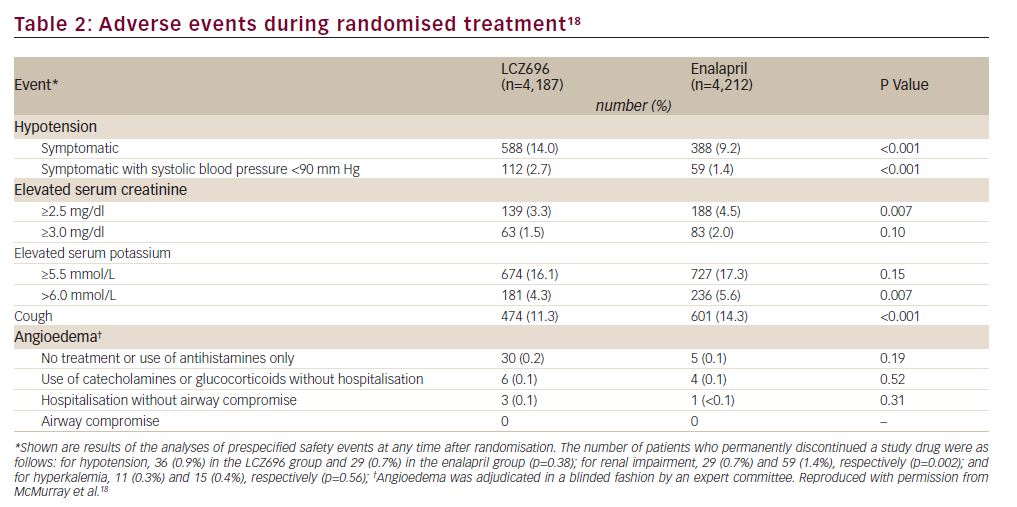
Overall, fewer patients stopped their study medication because of an adverse event in the LCZ696 group than in the enalapril group (10.7% versus 12.3%; p=0.03).17No major safety signals emerged for LCZ696 therapy.>sup>18The LCZ696 group had lower proportions of patients with renal impairment, hyperkalaemia and cough than the enalapril group, though the proportion of patients with hypotension was higher in the LCZ696 group (Table 2). Angioedema occurred very rarely and although it was more frequent in the LCZ696 than in the enalapril group, the study was not powered to show differences in such rare events (Table 2).
When comparing the results of the SOLVD-T and the CHARM-alternative trials with those of the PARADIGM-HF study it may be inferred that LCZ696 doubles the effect on cardiovascular mortality of renin angiotensin aldosterone system inhibitors.2,18,24Furthermore, an indirect comparison of LCZ696 with a putative placebo from SOLVD-T study25showed that LCZ696 may induce a RRR of 34% (95% CI: 21–44%; p<0.0001) in cardiovascular death, of 28% (95% CI: 16–39%; p<0.0001) in all-cause mortality and of 49% (95% CI: 39–58%; p<0.0001) in HF hospitalisation compared to placebo. These benefits were obtained even though LCZ696 was added to comprehensive background beta-blocker and MRA therapy.
Summary
PARADIGM-HF findings underscore the benefits of LCZ696 treatment versus enalapril for patients with HFrEF, with strong evidence based on: a rigorous trial design, large sample size, clinically meaningful endpoint, and a powerful beneficial effect with a very small p-value indicating high statistical significance. Further, a consistent effect was observed across a wide range of studied subgroups and benefits were achieved even though LCZ696 was added to comprehensive background BB and MRA therapy. Patients in PARADIGM-HF had similar rates of implantable cardioverterdefibrillator (ICD) device use as in other contemporary trials such as EMPHASIS-HF6 and RED-HF26and more than in SHIFT trial.27
More patients with HFrEF will soon be able to benefit from this new treatment. LCZ696 [sacubitril/valsartan tablets] was approved by the US Food and Drug Administration (FDA) for the treatment of HFrEF in July 2015.28Further, the agent is approved in the EU as well as in Switzerland. In addition, the 2014 Canadian Cardiovascular Society Heart Failure Management Guidelines Focus Update recommended that in patients with mild to moderate HF, an EF<40%, an elevated NP level or hospitalisation for HF in the past 12 months, a serum potassium level below 5.2 mmol/L and an estimated glomerular filtration rate of 30 mL/min or greater and treated with appropriate doses of guidelinedirected medical therapy should be treated with LCZ696 in place of an ACEI or an ARB.29It will be of interest to discover what approach will be adopted in future European and US guidelines.
CHARM – Candesartan in Heart Failure-Assessment of Reduction in Mortality and Morbidity
CONSENSUS – Effects of enalapril on mortality in severe congestive heart failure, results of the cooperative north Scandinavian enalapril survival
EMPHASIS-HF – The Eplerenone in Mild Patients Hospitalization and Survival Study in Heart Failure
PARADIGM-HF – Prospective Comparison of ARNI with ACEI to Determine Impact on Global Mortality and Morbidity in Heart Failure
RED-HF – Reduction of Events With Darbepoetin Alfa in Heart Failure
SHIFT – Systolic Heart failure treatment with the if inhibitor ivabradine Trial
SOLVD – Studies of Left Ventricular Dysfunction