The American Diabetes Association (ADA) defines prediabetes as glycated haemoglobin (HbA1c) 5.7–6.4% or fasting plasma glucose (FPG) 100–125 mg/dL or 5.6–6.9 mmol/L.1 Patients with prediabetes have up to a 70% chance of developing diabetes and a two-fold higher risk of cardiovascular disease than normoglycaemic patients. Studies show results varying from no association to a strong association between prediabetes and major adverse cardiovascular events (MACE) following percutaneous coronary intervention (PCI).2–4 Choi et al. reported a higher incidence of coronary restenosis and mortality in the prediabetic cohort after PCI compared with patients with normoglycaemia.5 Retrospective subgroup analysis of two randomized controlled trials of drug-eluting stents (DES) showed higher cardiovascular mortality in prediabetic patients versus normoglycaemic patients, but no difference in bleeding rates.6 Another interesting analysis depicted higher mortality with both low (<5.5%) and high (>8.0%) HbA1c among patients admitted for PCI.7 The results were compared with the reference group, whose HbA1c ranged from 6.1% to 7.0%; which represented the fraction of the reference group that met criterion for prediabetes and had better outcomes. Owing to contradictory literature, the PCI outcomes of prediabetic patients remains debatable. We performed a meta-analysis of 17 studies to better understand the outcomes of PCI across the spectrum of glycaemic control, i.e. normal glucose metabolism, prediabetes and diabetes mellitus (DM).
Methods
We conducted a systematic review and meta-analysis according to Cochrane collaboration guidelines and reported the results using the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (Figure 1).8 We searched the PubMed, Embase, Cochrane and Google Scholar databases using the terms “prediabetes”, “diabetes mellitus”, “percutaneous coronary intervention” or “PCI”, “ischaemic heart disease”, and “coronary artery disease” from inception until June 2022 without any language restrictions. All relevant publications, review articles and their references were manually screened to retrieve additional eligible studies.
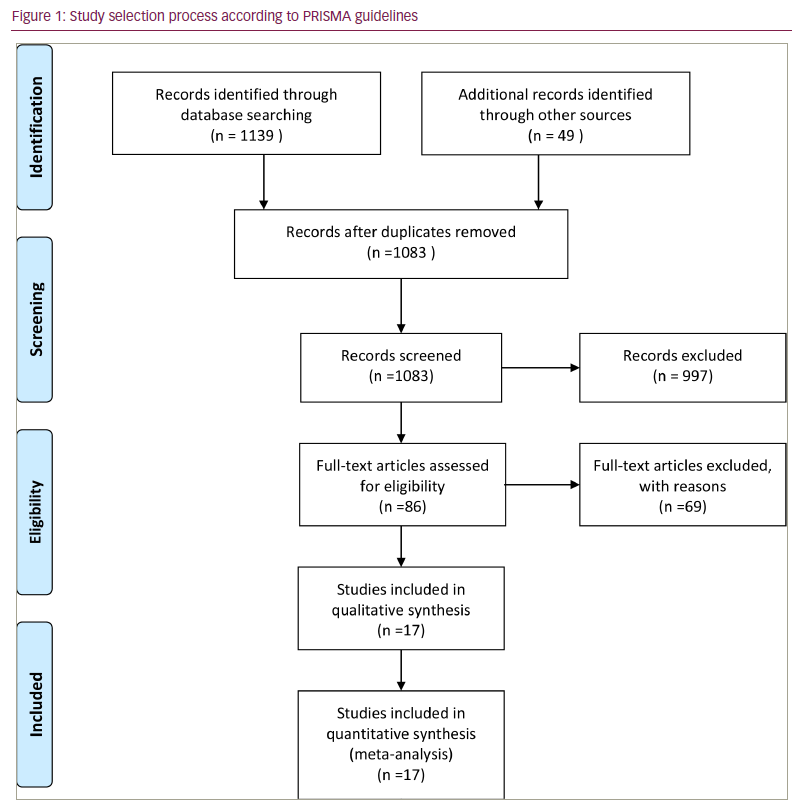
We included only full manuscripts of studies that met the following inclusion criteria: 1) compared patients with prediabetes and normoglycaemia undergoing PCI; 2) compared patients with prediabetes and DM in patients undergoing PCI. Prediabetes and DM were defined according to the ADA (2021) definitions:9 prediabetes was defined as HbA1c 5.7–6.4%, FPG 100–125 mg/dL, or an oral glucose tolerance test 2 hour plasma glucose 140–199 mg/dL; DM was defined as HbA1c ≥6.5%, FPG ≥126 mg/dL (7 mmol/L) or 2 hour plasma glucose ≥200 mg/dL (11.1 mmol/L). The following studies were excluded: 1) duplicates of previous publications; 2) studies reporting the same patient population as another included study; 3) studies without data on PCI outcomes; 4) studies reporting in-hospital outcomes only; 5) studies comparing PCI outcomes of normoglycaemic or diabetic patients only; 6) abstracts, editorials, reviews and commentaries; 7) animal studies.
The primary outcome of interest was all-cause mortality, and secondary outcomes were myocardial infarction (MI), cardiac death, target-vessel revascularization (TVR), target-lesion revascularization, stent thrombosis and stroke. Two reviewers independently extracted data from the eligible studies using a standardized data-collection form. The quality of the included studies was assessed using the Newcastle-Ottawa Scale. Any discrepancies regarding extracted data by the two reviewers were resolved by discussion among all the authors.
For all outcomes in our analyses, pooled risk ratios (RR) with their corresponding 95% confidence intervals (CIs) were calculated using the Mantel-Haenszel random-effects model for dichotomous variables. The Z-test was used to determine the significance of the pooled RRs. Heterogeneity across the studies was assessed using the chi-square-based Cochran’s Q test, and quantified using Higgins and Thompson’s I2 statistics. A Cochran Q statistic with a p-value ≤0.05 was considered significant. I2 statistic values of 25%, 50% and 75% were used to define low, moderate and high heterogeneity, respectively. Finally, we constructed funnel plots to assess for potential publication bias by plotting the standard error against the log RR (Suppl File 1). The meta-analysis was performed using Review Manager (RevMan) Version 5.3 (The Nordic Cochrane Center, Cochran Collaboration, Copenhagen, Denmark). Lastly, meta-regression analysis was performed using STATA 17.0 (StataCorp, College Station, TX, USA) to measure the influence of DES use on all-cause mortality and MI. A p-value of <0.05 was considered significant for all pooled analyses.
Results
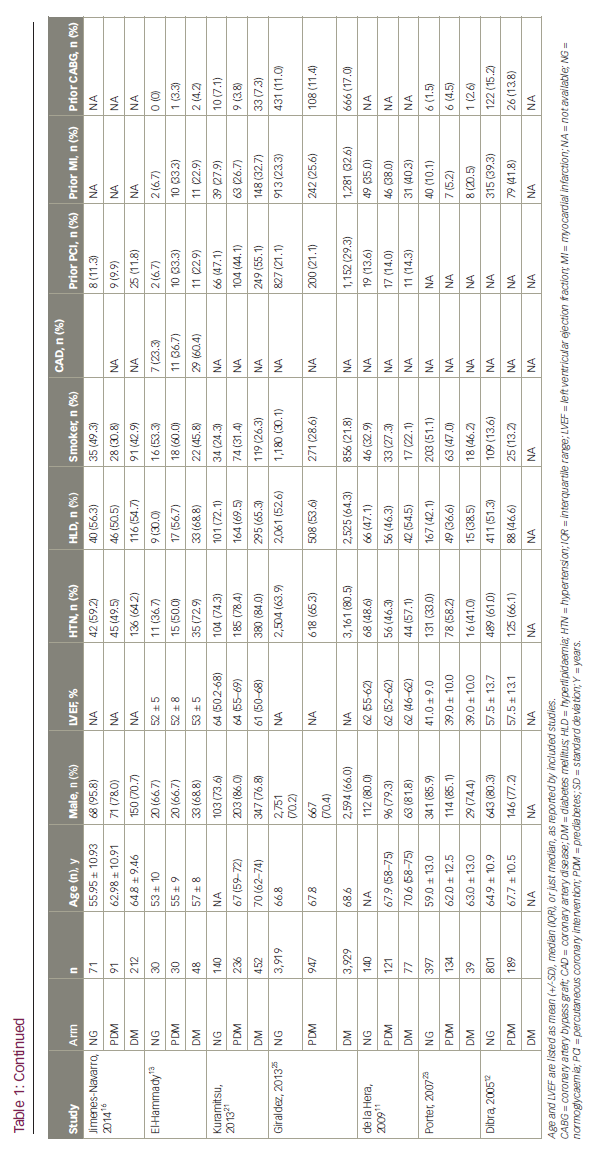
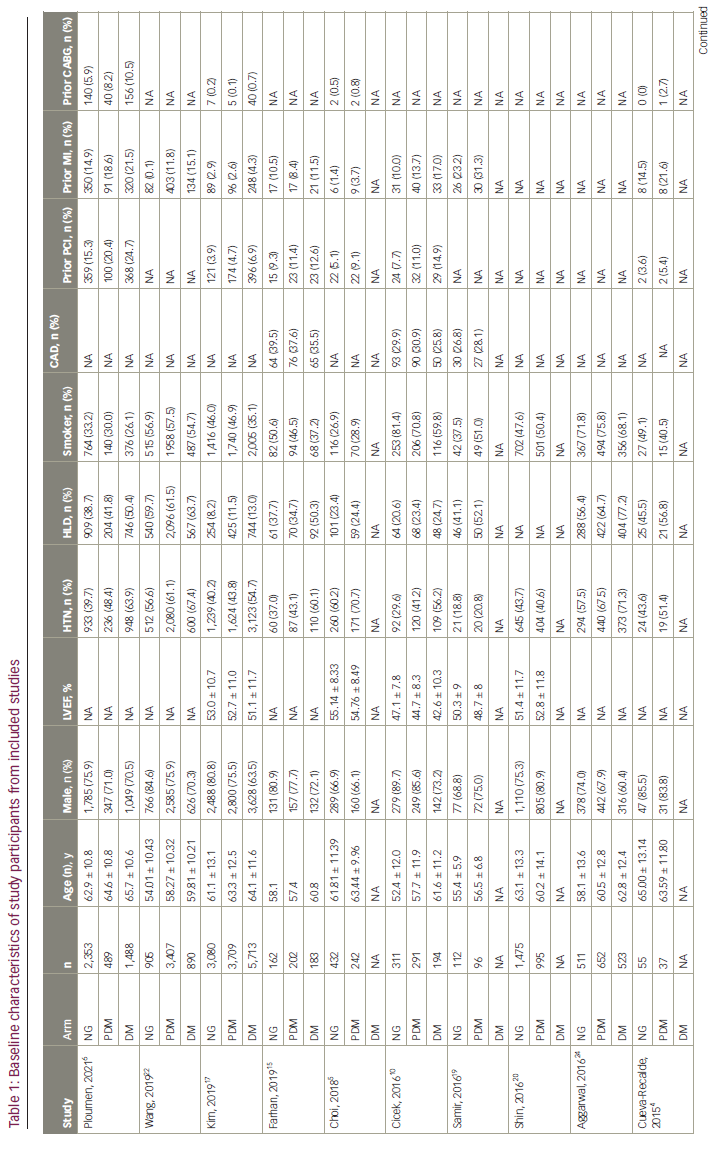
Of the 1,139 studies initially identified in the search, 17 studies3,5,10–23 (12 prospective and five retrospective) with 11,868 (76.5% male), 14,894 (78.5% male) and 13,536 (71.1% male) patients in the prediabetes, normoglycaemia and DM groups, respectively, were included in the final analysis. Seventeen studies compared outcomes for prediabetes versus normoglycaemia, while 12 studies compared outcomes for prediabetes versus DM at the longest follow-up. The mean follow-up duration was 2.6 years. Mean age (standard deviation) in each group was 62.1 (± 10.6) years, 60.2 (± 11.4) years and 69.0 (± 9.6) years, respectively. Table 1 summarizes baseline patient characteristics. A summary of study characteristics, definitions of prediabetes and DM, and PCI indications are included in Suppl Table 1.
Details about target vessel and use of DES stratified by prediabetes status across the studies are shown in Suppl Table 2. Discharge medications are outlined in Suppl Table 3.
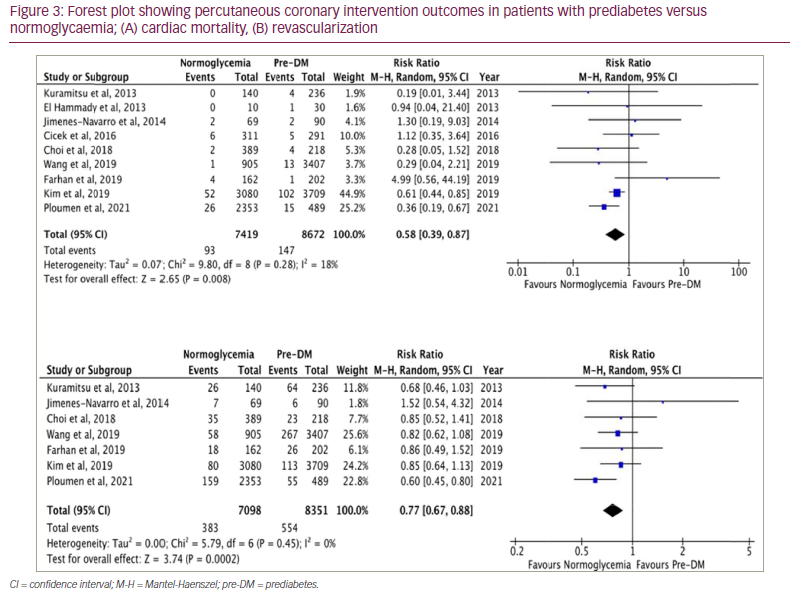
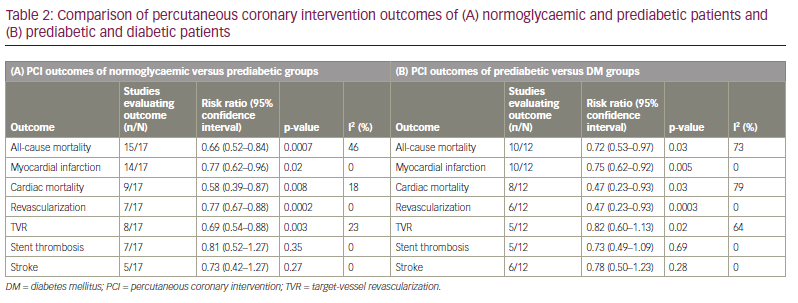
Among patients undergoing PCI, the normoglycaemic group had lower risk of all-cause mortality, MI, cardiac mortality, revascularization and TVR compared with the prediabetic group (Figures 2 and 3, Suppl Figure 1). There was no difference between the prediabetes and normoglycaemia groups for post-PCI stent thrombosis and stroke. The findings are outlined in Table 2(A).
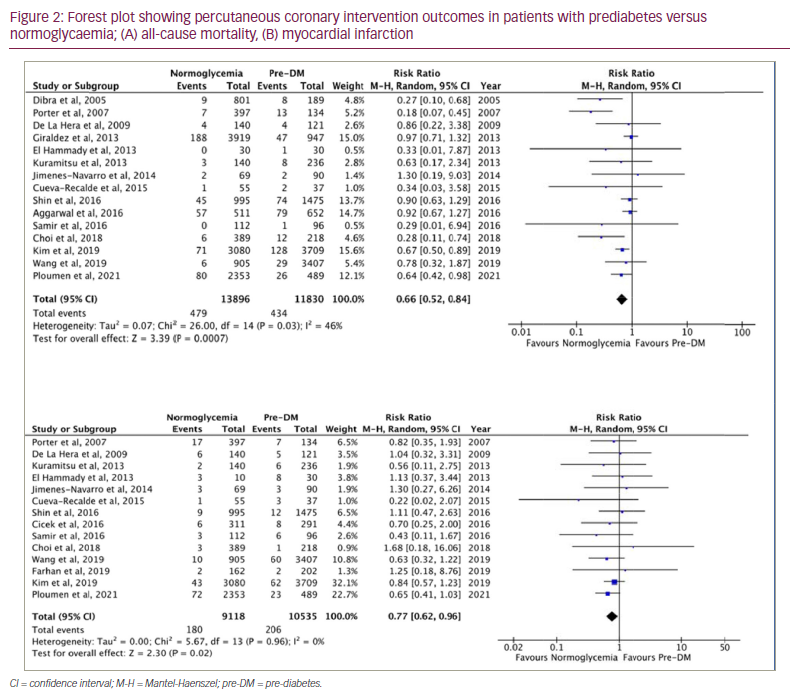
In the subgroup analysis for patients included in prospective studies, normoglycaemic patients who underwent PCI had lower all-cause mortality (RR=0.71 [95% CI 0.51–0.99]; p=0.04; I2=44%), MI (RR=0.74 [95% CI 0.57–0.97]; p=0.05; I2=0%) and TVR (RR=0.66 [95% CI 0.48–0.90]; p=0.009; I2=39%) compared with patients in the prediabetes group, while there was no difference in cardiac death (RR=0.76 [95% CI 0.33–1.71]; p=0.50; I2=41%) between the two groups. Similarly, subgroup analysis of prospective studies showed that patients in the prediabetes group had lower all-cause mortality (RR=0.64 [95% CI 0.51–0.80]; p<0.0001; I2=0%), MI (RR=0.75 [95% CI 0.62–0.92]; p=0.05; I2=0%) and cardiac death (RR=0.47 [95% CI 0.23–0.93]; p=0.03; I2=80%) compared with the DM group, with no difference in TVR (RR=0.88 [95% CI 0.59–1.33]; p=0.55; I2=70%) between groups (Suppl Figures 3–6).
Our meta-regression analysis showed that DES use during PCI was associated with a significant increase in the incidence of MI in the prediabetes group when compared with DM groups (p=0.048). However, DES use did not affect the incidence of MI in patients with prediabetes versus normoglycaemia (p=0.80). Similarly, there was no significant association between DES use and the incidence of all-cause mortality in patients with prediabetes versus normoglycaemia (p=0.36) or prediabetes versus DM (p=0.52) (Suppl File 2).
Discussion
In this meta-analysis, we evaluated the impact of the degree of dysglycaemia on cardiovascular morbidity and mortality in patients undergoing PCI. Our findings suggest that, when compared with normoglycaemic patients, prediabetic patients undergoing PCI had higher risk of all-cause mortality, MI and revascularization, with no difference in risk of post-PCI stent thrombosis and stroke. Compared with patients with DM, patients with prediabetes undergoing PCI had lower risk of all-cause mortality, cardiac mortality, recurrent MI and revascularization, with no difference in stent thrombosis or stroke between the two groups.
Multiple studies have reported prediabetes to be associated with an increased risk of mortality in the general population and in patients with CAD. Although data about the risk of mortality in prediabetic patients undergoing PCI are inconsistent, our study reports that these patients are at increased risk of all-cause mortality in this subgroup of CAD patients compared with normoglycaemic patients. These findings are consistent with the meta-analysis of Cai et al. that analysed 129 studies and reported that prediabetes was associated with an increased risk of mortality in the general population and in patients with atherosclerotic cardiovascular disease.26 Another recent meta-analysis of 12 studies showed that prediabetes is an independent prognostic factor of MACE after PCI.2 It highlighted that prediabetic patients undergoing PCI have a higher risk of adverse outcomes compared with normoglycaemic patients. However, it did not compare the outcomes of prediabetic patients with those of patients with DM. Our meta-analysis fills in the gap to understand the impact of glycaemic control on PCI outcomes for CAD.
Although prediabetic patients undergoing PCI have an increased risk of mortality compared with normoglycaemic patients, this risk was lower compared with patients with DM. Our findings are similar to those reported in the analysis by Zhong et al. who found that the curves for mortality were relatively flat when HbA1c levels were less than approximately 5.7%, and rose steeply thereafter.27 These findings can be explained by the fact that cardiovascular risk factors begin to impact the patient long before the diagnosis of DM. The duration of impaired glucose tolerance (IGT) not only influences CAD risk, but is also associated with insulin resistance. IGT contributes to a spectrum of risk factors that contribute to the development of metabolic syndrome, thus increasing the risk of CAD long before the onset of DM.28,29 Secondly, this can partly be due to the fact that when prediabetic patients present with acute MI, they often receive less-aggressive treatment than those with DM, as prediabetes is perceived by the treating physicians to be a less aggressive disease requiring only lifestyle modifications and exercise.30 Thus, these findings support screening for abnormal glycaemic metabolism in CAD patients undergoing PCI, and treating patients with impaired glucose metabolism aggressively with antidiabetic medications with cardiovascular benefits, such as sodium–glucose co-transporter-2 inhibitors31–34 and glucagon-like peptide-1 receptor agonists.35,36 Since prediabetes is an established risk factor for MACE, lowering HbA1c can potentially have a preventive value.37
Prediabetes has been attributed to an increased risk of MACE in patients with CAD.38 Our study showed that prediabetic patients undergoing PCI had a higher incidence of recurrent MI and revascularization compared with normoglycaemic patients, and that these risks were lower compared with patients with DM. These findings are relatable to the study by Kim et al. that reported that prediabetes could have a similar impact to DM on major clinical outcomes in patients with ST-elevation MI and multi-vessel disease.39 A likely explanation of these findings is the pathophysiological mechanism: prediabetes is associated with systemic inflammation, insulin resistance and production of reactive oxygen species by hyperglycaemia, which leads to endothelial dysfunction, impaired microvascular function, increased prevalence of multi-vessel disease, and likely progression to DM over time.40 Amano et al. confirmed this hypothesis and performed an intravascular ultrasound study, which showed that patients with IGT were more likely to have lipid-rich coronary plaque as compared with normoglycaemic patients.41 Similarly, Ertan et al. also reported patients with prediabetes had smaller coronary size and diffuse coronary narrowing compared with normoglycaemic patients, which may increase the risk of adverse cardiac events like MI, and the need for revascularization after PCI.42
Although DM has been associated with increased incidence of stent thrombosis after PCI,43 our study reported there was no difference in the incidence of post-PCI stent thrombosis between the prediabetic versus normoglycaemic groups, and prediabetic versus DM groups. Similarly, we reported there was no difference in risk of stroke between the prediabetic versus normoglycaemic groups, and prediabetic versus DM groups. This is similar to findings of Mitsios et al., who reported that there was no difference in the risk of first stroke when patients with normoglycaemia and prediabetes were compared.44 However, this contrasts with the findings of a meta-analysis by Lee et al., who reported that prediabetes was associated with a higher risk of stroke and stroke-related morbidity, but the relative risks were modest, associated with significant heterogeneity and not consistent when different definitions of prediabetes were used.45
There are several important limitations of our meta-analysis. The definitions of prediabetes and DM across studies were based on different criteria and, due to this, only a limited number of studies were available for comparison. Therefore, we were not able to perform a meta-analysis according to varying definition criteria of prediabetes/DM separately. Although the included studies did not all use the same criteria, each study met one of the three ADA-specified criteria for prediabetes/DM.9 Similarly, we did not include metabolic syndrome, as the definition varies from study to study. Additionally, although patients with prediabetes are more likely to progress to DM than those with normoglycaemia, most of the included studies did not adjust for progression to DM. However, the mean follow-up duration of 2.8 years was not long enough to attribute all the associated increased risk of mortality from progression of prediabetes to DM. Finally, no details about treatments were available for post-PCI patients in the included studies, so the effect of treatment on outcomes post-PCI cannot be evaluated.
Conclusions
Among CAD patients who underwent PCI, the risk of all-cause and cardiac mortality, MI and revascularization in prediabetic patients was higher compared with normoglycaemic patients, but lower compared with patients with DM. Thus, patients undergoing PCI should be screened for prediabetes and treated optimally.