Aortic stenosis is a common degenerative valve disease and its prevalence increases with age.1 If untreated, severe symptomatic aortic stenosis leads to significant morbidity and mortality.2 Although surgical aortic valve replacement (SAVR) is a well-established treatment option for this condition, more than 30% of patients are not suitable candidates for SAVR due to increased operative risks; advanced age and age-related frailty; left ventricular dysfunction; and multiple comorbidities, including pulmonary and renal dysfunction.3,4 Transcatheter aortic valve replacement (TAVR), a percutaneous, minimally-invasive, cathlab-based procedure, was first performed by Alan Cribier et al. in 2002, and has revolutionised the treatment of calcific aortic stenosis in the elderly.5 With increasing favourable evidence from registries and randomised trials versus SAVR, TAVR has now become the new standard of care for elderly patients with calcific aortic stenosis.6–9
Although TAVR started as an alternative treatment for patients with severe aortic stenosis who were deemed inoperable or at high-risk for SAVR,10,11 it has now shown advantages and superiority to SAVR even in low surgical-risk patients.12 With rapidly increasing numbers of patients and widening indications, there is an unmet need to further improve TAVR technology in order to reduce the procedural risks of conduction disturbance, paravalvular leak, stroke and vascular complications, making the procedure safer for the patient and less complex for the operator.
The Myval™ transcatheter heart valve (THV) (Meril Life Sciences Pvt. Ltd., Gujarat, India) is a CE-marked, newer-generation balloon-expandable TAVR system. After initial animal studies and early data from the first-in-human MyVal-1 study, which demonstrated safety and effectiveness of the Myval THV for the treatment of severe aortic stenosis in patients at intermediate or high-risk for surgery,13 the Myval THV was approved by the Drug Controller General of India (DCGI) in October 2018. To date, more than 1,800 patients worldwide have been treated with the Myval THV. This review summarises the salient technical features of the Myval THV, clinical experience and the published and ongoing trials and registries.
Technical features of the Myval transcatheter heart valve system
The Myval THV system is made up of three main components: the valve, the delivery catheter and the introducer sheath.
The valve
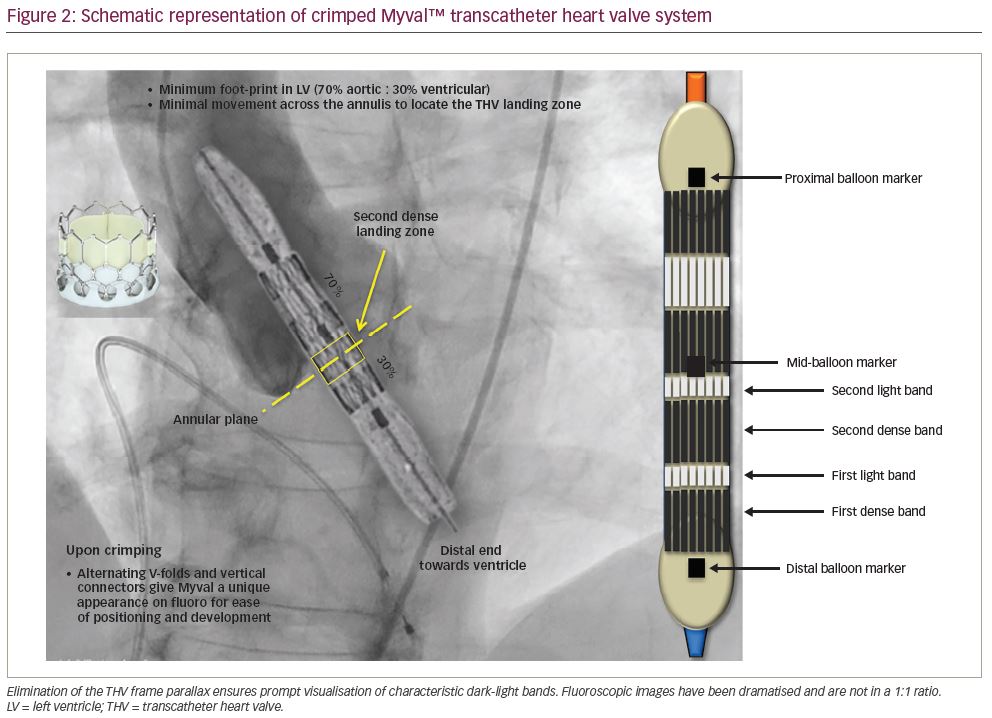
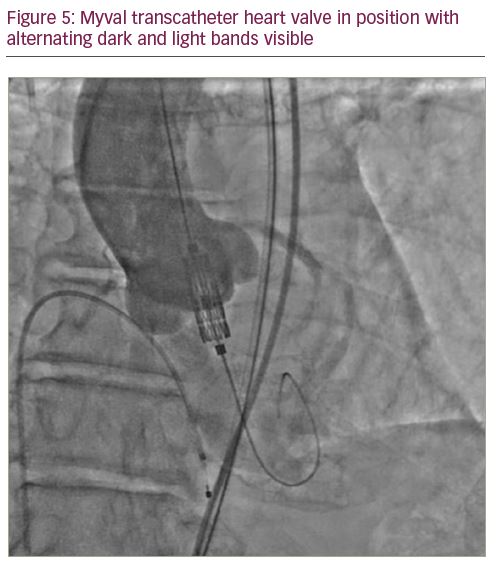
The valve consists of a tri-leaflet, decellularised, bovine pericardial valve treated with anti-calcification treatment and fixed at three equipoise vertical commissural posts (separated by 120 degrees) on the metal frame. The nickel–cobalt alloy stent frame is composed of a single design element – hexagons, arranged in such a fashion that allows large open cells to occupy 53% of the frame towards the aortic end and closed cells to occupy 47% of the frame towards the ventricular end (Figure 1). The open cells in the upper half ensure optimal perfusion of the coronary arteries, whereas the closed cells in the lower half provide high radial strength. The valve frame, upon crimping under fluoroscopy, gives rise to a unique dark–light alternating band-like pattern (attributed to the novel honeycomb hybrid design). The second dark band from the ventricular end serves as the marker facilitating precise positioning and deployment at the desired location (Figure 2). The lower closed cells of the valve frame are covered externally with a protective sealing cuff of polyethylene terephthalate to form an external buffing, which provides a protective sealing cuff to reduce, or nearly eliminate, paravalvular leak.
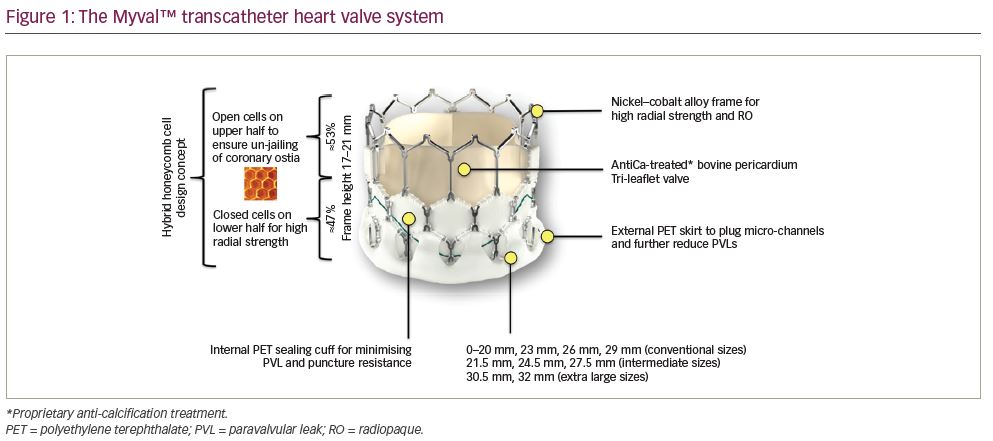
The Myval THV is available in various sizes – conventional (20.0, 23.0, 26.0 and 29.0 mm), intermediate (21.5, 24.5 and 27.5 mm) and extra large (30.5 and 32.0 mm). It is worth noting that all sizes of Myval THV are approved by the Central Drugs Standard Control Organisation, India and are CE-marked. The availability of intermediate sizes facilitates precise sizing to match the annulus, thereby reducing oversizing risks. Also, the availability of the extremely small and large sizes enables the treatment of a range of aortic annulus diameters from 18.5 mm to 29.9 mm with standard and intermediate Myval THV sizes. The treatable range further expands to 32.7 mm with the extra large size of Myval THV.
The delivery catheter
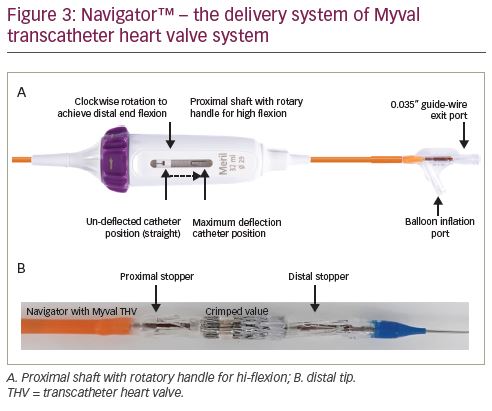
The valve delivery system, named Navigator (Meril Life Sciences Pvt. Ltd., Gujarat, India), (Figure 3) has a unique design characterised by a proximal deep flexion handle and a distal over-the-wire balloon, on which the Myval THV is pre-mounted externally. External crimping and mounting on the balloon simplifies the procedure for the operator. The Navigator system on either end has two counter-opposing soft stoppers that create a shallow, low-profile crimping zone to provide a precise and snug fit of the crimped valve. These stoppers prevent inadvertent migration of the valve and minimise the risk of valve dislodgement during its advancement through the sheath and the aorta. The Navigator system has a high-flexion feature which allows flexion of the distal catheter system and reduces the risk of trauma to the aortic arch during advancement, and thereby possibly reduces the risk of peri-procedural stroke. Additionally, this flexion feature facilitates crossing a difficult and angulated or horizontal annulus. Another important feature of the balloon is that it has two internal expansion ports, which facilitates simultaneous expansion, distally and proximally (like a dog bone), which stabilises the valve during deployment and ensures precise placement.
The introducer sheath
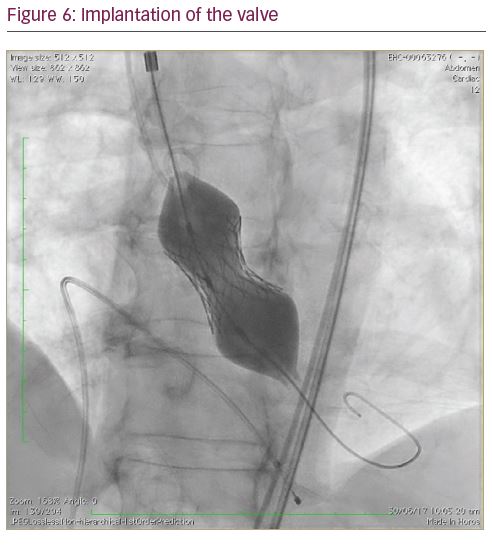
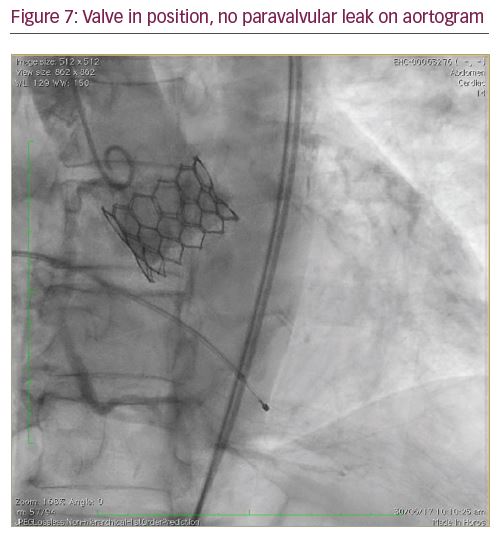
The Python™ introducer sheath (Meril Life Sciences Pvt. Ltd., Gujarat, India) is a 14 French sheath which expands momentarily (like a python swallowing prey) to allow passage of the Myval THV crimped on balloon catheter. Two separate, calibrated loading tubes ensure the temporary opening of haemostatic valves in the proximal port, allowing smooth passage of the crimped Myval THV System. The unique feature of this sheath allows the complete retrieval of an undeployed Myval THV in the event of a difficult crossing or inadvertent loss of the LV wire position, and facilitates insertion of it again through the sheath (Figures 4–7).
The Myval THV is typically designed to be delivered through the transfemoral approach; however, trans-subclavian, transaortic, and trans-carotid approaches have also been used.
Pre-clinical studies
Initial animal studies were conducted as per standard protocols. Successful aortic anchoring, along with acute valve functionality of the Myval THV was achieved in all the ovine TAVI aortic banding models.14 During the pre-clinical tests, Myval THVs were successfully implanted in sheep via the transcarotid approach. Notably, there was no device-related mortality, and transthoracic echocardiography of the surviving sheep showed that all valves were functional with no significant regurgitation or calcification.15 There was no evidence of thrombus/embolisation or structural abnormality in the valve and its components.
Clinical experience with the Myval transcatheter heart valve system
The first-in-human MyVal-1 study (Table 1) was a prospective, multicentre, single-arm, open-label study, conducted at 14 clinical sites across India.13 The study assessed the safety and effectiveness of the Myval THV in 30 patients with severe aortic stenosis who were classed as intermediate or high-risk for surgery. Clinical follow up and echocardiography were performed immediately post-procedure and at 1, 6 and 12 months following the procedure. The safety endpoint was Kaplan–Meier survival at 12 months follow up. Efficacy endpoints, as defined in Valve Academic Research Consortium-2 (VARC-2),16 included improvement in New York Heart Association (NYHA) functional classification, (effective orifice area) EOA, and 6-minute walk test from baseline and 12 months follow up. The study also determined quality of life, as measured by the Kansas City Cardiomyopathy Questionnaire.
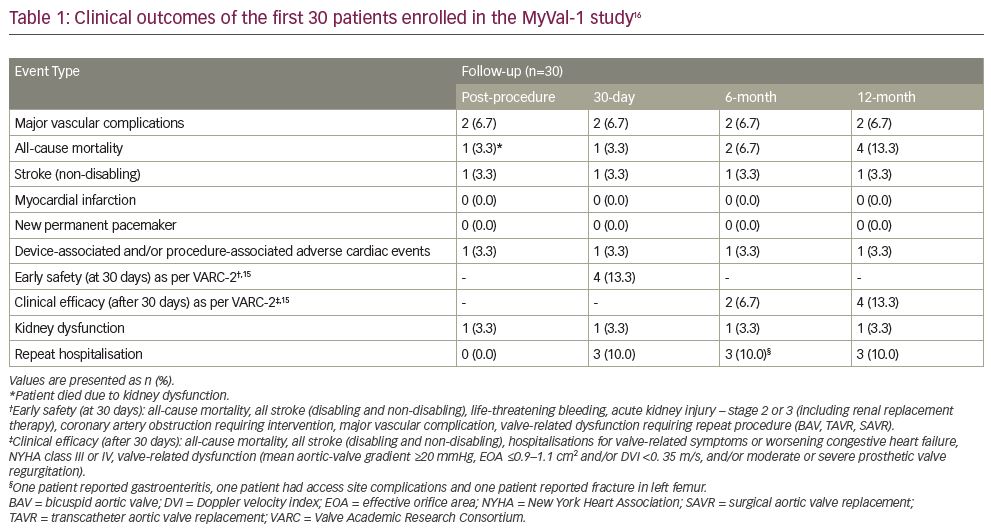
The mean age of patients was 75.5 ± 6.7 years, with mean Society of Thoracic Surgeons score of 6.4 ± 1.8%. It is noteworthy that 70% of patients had NYHA functional class III/IV. Post-procedural echocardiography showed significant improvement of EOA (1.7 ± 0.3 cm2 versus 0.6 ± 0.2 cm2, p<0.0001) and mean aortic-valve gradient (8.0 ± 2.7 mmHg versus 47.4 ± 8.8 mmHg, p<0.0001), compared with pre-procedure. Haemodynamic performance of the valve sustained at 12 months, with EOA of 1.8 ± 0.3 cm2, peak aortic-valve gradient of 20.3 ± 5.9 mmHg, and mean aortic-valve gradient of 12.0 ± 3.3 mmHg.
At 12 months clinical follow up, reported all-cause mortality was four patients (13.3%). Of the four all-cause mortality cases, one patient died due to a vascular complication leading to acute renal failure post-procedure, one patient died due to septicaemia at 6 months follow up, one patient died due to coronary artery disease with hypertension, and death related to a non-cardiac event was reported in another patient at 12 months follow up. Major vascular complications were observed in two patients post procedure: one non-disabling stroke; no myocardial infarction, haemolysis, thrombosis, or valve migration was reported in any of the patients. None of the patients required a new permanent pacemaker at 12 months follow up. NYHA functional class improvement was noted in all the patients at 12 months follow up. There was also a substantial improvement in quality of life (36.6 ± 11.0 versus 65.9 ± 11.4) and the 6-minute walk test (148.0 ± 87.4 versus 336.0 ± 202.9 m) from baseline to 12 months follow up.
The MyVal-1 study was further extended to include 100 patients from over 30 sites in India (Table 2).17 At 6 months follow up, 6-minute walk test and Kansas City Cardiomyopathy Questionnaire scores were improved compared with baseline.17 The overall 6-month all-cause mortality was 9% and stroke was 2%. The rate of new permanent pacemaker implantation was 2% (one patient had right bundle branch block pre-procedure). There was no case of myocardial infarction at 6 months follow up. Patients’ hospital visits were delayed due to the COVID-19 pandemic and therefore, the echocardiographic follow up is still ongoing.
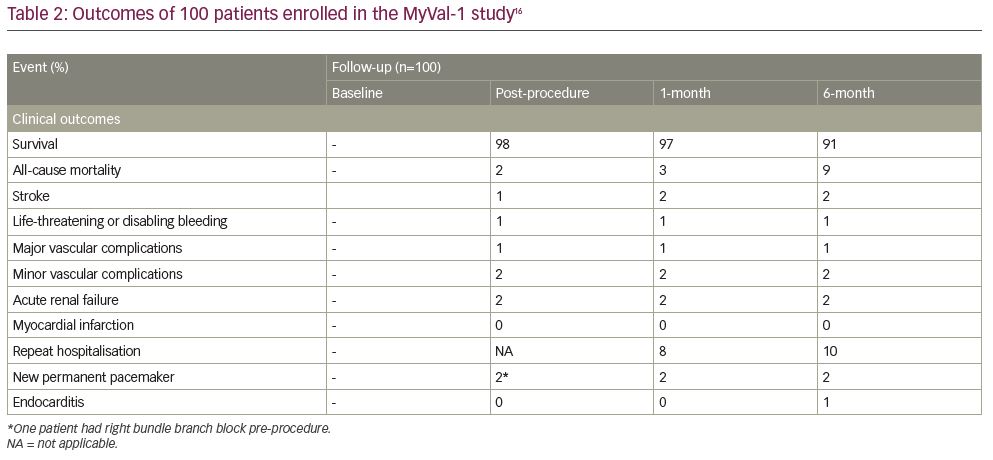
The results of MyVal-1 study demonstrated the primary safety and effectiveness of the Myval THV at 6 months post-procedure. With more than 1,800 implants across the globe, experience from unpublished registries as well as our own experience have shown successful results with ease of deployment in a wide range of complex anatomies, including bicuspid aortic valves with low complication rates.
Future directions
With very promising initial results, the Myval THV system now needs to be studied in a larger patient population involving long-term follow up. To achieve this, a pivotal randomised trial, the LANDMARK TRIAL (ClinicalTrials.gov Identifier: NCT04275726), is planned, which will compare the Myval THV to the most commonly used THV systems. The ongoing comparison of the Myval THV with SAPIEN 3 (Edwards Lifesciences, Irvine, CA, USA) is being studied in the MATCH-BALL trial (ClinicalTrials.gov Identifier: NCT04548726), under the hypothesis that there are differences in terms of transvalvular gradients and residual paravalvular leak amongst different balloon-expandable TAVR devices available in the market. The aim of the MATCH-BALL trial is to compare the haemodynamic performance of these two balloon-expandable TAVR devices.
LANDMARK is a prospective, randomised, controlled, non-inferiority trial, designed to compare the Myval THV with newer-generation contemporary valves (SAPIEN THV series and Evolut™ THV series [Medtronic, Dublin, Ireland]). The trial is expected to enrol 768 patients with severe symptomatic native aortic valve stenosis. Patients will be randomised in a 1:1 ratio to receive either the Myval THV or contemporary valves (SAPIEN THV series or Evolut THV series). The primary endpoint of the trial is a combined safety and effectiveness, which is a composite of all-cause mortality, stroke, life-threatening/disabling bleeding complications, major vascular complications, acute kidney injury, moderate/severe prosthetic valve regurgitation and requirement of new permanent pacemaker implantation at 30 days follow up. The trial was due to start in the second quarter of 2020, but has been delayed because of the COVID-19 pandemic. Ethical approval has been granted for many centres and the initial lead-in cases have been performed at a few centres. Randomisation is expected to begin at the earliest available opportunity.
Another real-world registry, which is well on the way is the Myval European study (ClinicalTrials.gov Identifier: NCT04703699; n=200 patients from approximately 15 sites across Europe). This registry hopes to shed light on the safety and efficacy of Myval THV in contemporary clinical practice.
Conclusion
TAVR has become the standard of care for patients with symptomatic severe aortic stenosis. Continuous improvements in technology over the earlier generations of THVs have led to improved efficacy and safety, making this technology available to wider population. The Myval THV is a newer-generation, CE-marked balloon-expandable valve, consisting of tri-leaflet bovine pericardial leaflets, supported by a nickel–cobalt alloy frame, delivered through a high-flexion Navigator balloon catheter system. Early results from the MyVal-1 first-in-human trial are promising, with excellent procedural success, precise deployment and good outcomes in short-term follow up. Longer-term follow up and ongoing randomised controlled trials and real-world registries with the Myval THV are expected to further support the safety and efficacy, and to expand its indication across the world.