Background: Ripple mapping (RM) displays the entire morphology of bipolar electrograms in 3D, facilitating identification of low amplitude pathways often obscured by large far-field signals. We tested the hypothesis that RM improves characterisation of low amplitude signals in three subgroups where such signals are common: atrial tachycardia, the LV conduction system and post infarct LV scar.
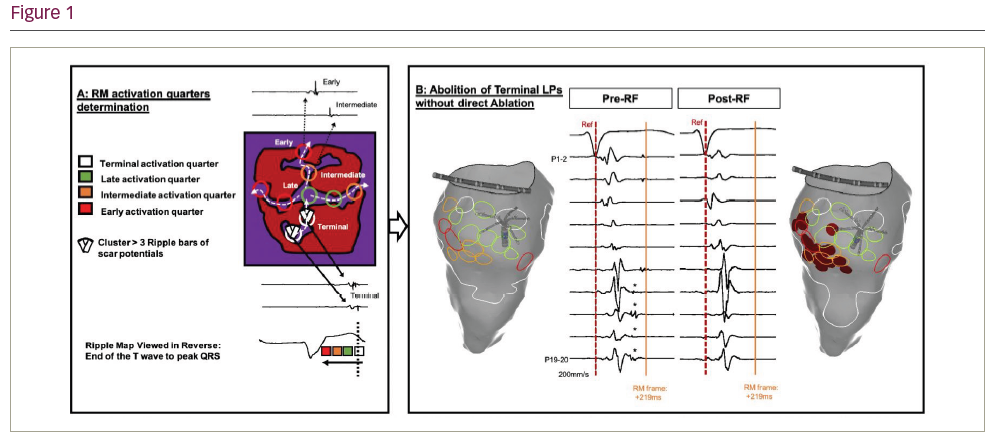
Methods and Results: Scar-related atrial signals: We reviewed 28 cases of perimitral AT, ablated with either RM or conventional LAT mapping. A slow conducting scar-related isthmus was identified and ablated in 12 pts, all using RM to distinguish non-activating scar from low-voltage signals. Conventional mitral isthmus (MI) linear ablation was performed in 16 pts. Scar-related isthmus ablation terminated AT in 100% vs 63% with MI linear lesions (p=0.027). Slow conducting isthmuses could be identified by applying RM in 63% of those who had MI lines. Isthmuses were narrow (8.9 ± 3.5 mm), slow (31.8 ± 5.5 cm/s), low voltage (0.11 ± 0.05 mV) tissues bordered by scar. Ventricular pre-systolic signals: High-density maps of the LV during SR (points: 3,086 ± 482) in 17 pts undergoing VT ablation map were reviewed using RM. Two distinct wavefronts, from split presystolic bipolar EGMs (PSP), could be seen on all RMs irrespective of QRS morphology. PSPs were grouped into left anterior fascicle (LAF) and left posterior fascicule (LPF). The LAF was significantly shorter than the LPF: 43.7 vs 67.9 mm (p=0.0006), but conduction speeds similar: 1.6 vs 1.4 cm/s (p=0.5). The LPF was significantly shorter in Pt with LBBB (67.2 vs 18.1 mm; p<0.0001). LPF length inversely correlated with QRS duration (r -0.59; p=0.013). Ventricular post-systolic signal: RM was applied prospectively to 11 pts with post-infarct VT. High-density mapping of LV scar (3050 ± 554.5 points) was performed during SR (n=7), or pacing (n=4). RM identified channels of slow conduction within the scar (4 anterior, 7 inferior) of all pts. Component low-amplitude scar potential (SPs) were further categorised temporally: early, intermediate, late, terminal SPs by reviewing the RM in reverse to limit far-field interpretation. SP timing ranged: 98.1 ± 60.5 ms to 214.8 ± 89.8 ms. Earliest SPs were present at the border, occupying 16.4% of scar, whilst latest SPs occupied 4.8% at the opposing border (in 45.5%) or core (in 54.5%). Analysis took 15 ± 10 min to locate channels and identify ablation targets. Selective ablation of early SPs (Figure 1) led to elimination of the terminal SPs in all (mean ablation 16.3 ± 11.1 min). We then tested this observation as the procedural endpoint for substrate modification of post infarct VT in 50 pts at 5 centres (NCT: NCT03997201; mean age 70.2 ± 7.1 years, mean EF 32.9 ± 9.5%). The primary endpoint was combined ICD therapy, VT recurrence and mortality at 1 year. The protocol was abandoned in 2 and the RM endpoint met in 37 (74%). Where the endpoint was met, VT was non-inducible in 91%, and 86% are free of VT at median 6.5 [1.2 – 12.0] months. Where the endpoint was not met, VT was non-inducible in 55%, and 80% are free of VT at 4.6 [1.2 – 7.8] months. 3 pts died without VT recurrence. On an ITT basis, 40/50 (80%) were free from the primary endpoint at median 5.7 [1.8 – 12.0] months.
Conclusion: RM was able to map low amplitude signals in both atria and ventricle. This enabled ablation to be directed at novel targets for peri-mitral AT and post-infarct VT. Prospective randomised trials are needed to determine if these endpoints are superior to current strategies.