Purpose: In April 2017, the British Heart Rhythm Society (BHRS) published standards for implantation and follow up (FU) of cardiac rhythm devices. This document highlights that permanent pacemakers should be implanted within 24 hours of bradycardic emergencies.
In 2014 the London Clinical Senate endorsed the arrhythmia triage pathway. This pathway allows London ambulance service (LAS) to triage high risk patients and take them directly to an arrhythmia centre. This included Barts Heart Centre (BHC) which provides emergency 24/7 pacemaker and ablation procedures for the central and East London area. We audited our emergency pacemaker implants for the year 2017 to determine areas of service improvement.
Methods: We conducted a retrospective audit, including all patients who underwent an “emergency” permanent pacemaker. We excluded patients who had incomplete data and patients needing pacing as a result of a complication to a concurrent procedure. For comparison a control group was collected of elective permanent pacemaker implants at BHC in 2017. We systematically reviewed each patient’s record to assess:
- lab time used;
- admission to treatment time
- length of hospital stay defined as admission to discharge from hospital;
- complications, defined as occurring within or after 30 days from implant;
- pacing dependency at 1-, 4- and 16-months post-implant. “dependent” was defined if their underlying rhythm remained to be CHB; and
- device upgrades were judged as patients who had undergone or planned to undergo an upgrade procedure.
Results:
- 186 emergency implants were performed, 88.8% were BHRS compliant (median 4 hours);
- 82.8% (n=154) were dual chamber pacemakers (DDD), 16.1% (n=30) were single-chamber pacemakers and 1.1% (n=2) were biventricular pacemakers. Over the follow-up period only one was upgraded from a DDD to an internal defibrillator (ICD). This patient received a DDD post myocardial infarction for CHB, then developed symptomatic sustained VT 6 months later and upgraded to a dual chamber ICD;
- 85.4% (n=159) were discharged home, 12.4% (n=23) were discharged to their local hospital and 2.2% (n=4) died during this hospital admission from other comorbidities including ischaemic stroke (n=2), end of life pathway (n=1) and end-stage COPD (n=1);
- median hospital stay was 28 hours. The main reasons for a delay in discharge was social care issues (n=16) and trauma caused by the incident leading to the admission (n=10);
- the median lab time for emergencies (n=186) and elective cases (n=258) were both 105 mins. The mean time for emergencies (110.3 mins) was longer than the mean for elective (108.1 mins) but insignificant (p value = 0.224);
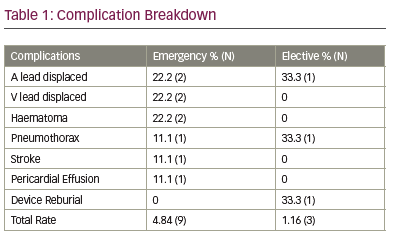
- emergency implants have a higher complication rate (p value = 0.009) see Table 1; and
- on average 65.4% of patients were deemed dependent at each follow-up interval.
Conclusion: Our results show that emergency implants during 2017 at the BHC were 88.8% BHRS compliant. Emergency implants carried a 4x increase of complications than elective pacemakers but procedure time was the same. Only 1 patient required an upgrade procedure after initial implant, suggesting that it was possible to appropriately select devices even with paramedic triage emergency pathways.