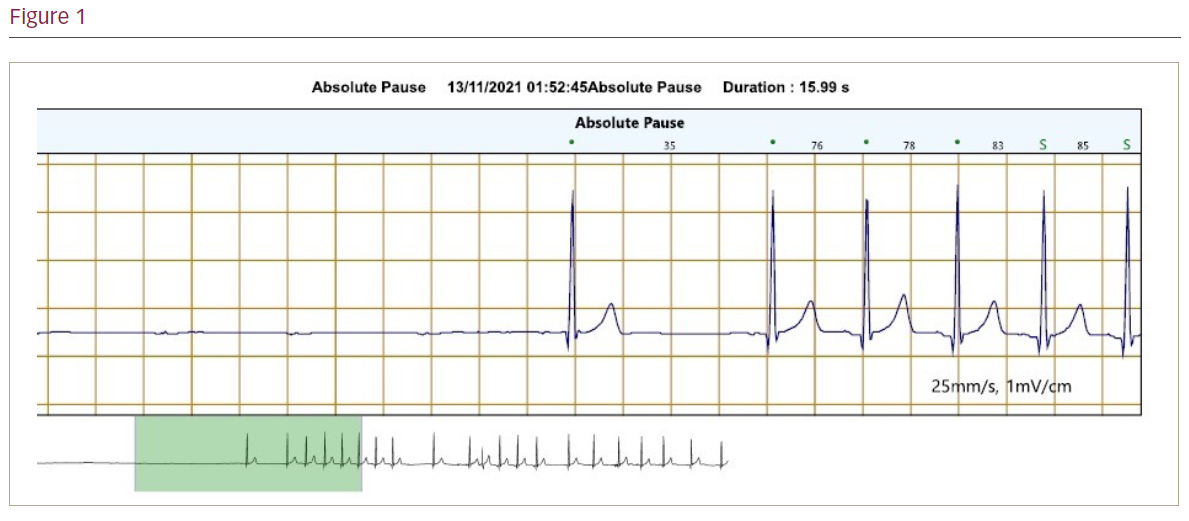
A 28-year-old man who worked as a scaffolder had been waking up in the middle of night gasping for air and with a sensation of chocking. His 7-day Holter revealed a 16-second pause (p wave asystole) associated with his symptoms (Figure). He had been experiencing such nocturnal symptoms sporadically for several years, having first been investigated with a 24-hour tape when he was 20 years old, which demonstrated nocturnal pauses up to 3.55 seconds and nocturnal second-degree atrioventricular block (2:1) lasting up to 13 seconds. An echocardiogram revealed no abnormality, and he had no other medical history though at the time he was a regular user of cannabis. He was lost to follow-up for several years until he was referred back to cardiology services following another presentation with palpitations waking him up at night. His body mass index was 26 and his Epworth score was 15, so he was referred for sleep studies for sleep apnoea work-up. His sleep study came back as negative for obstructive sleep apnoea (OSA). A repeat echocardiogram was also normal. He also had a cardiac magnetic resonance imaging scan, which showed mild left ventricular impairment and no evidence of scar tissue. Computed tomography coronary angiogram was negative for coronary artery disease. Other than his nocturnal symptoms he had no daytime bradyarrhythmias and no history of syncope, presyncope, chest pain or shortness of breath. Biochemical work-up and Lyme serology were negative. His case was taken to electrophysiology multidisciplinary team following the 16-second pause to discuss his young age, occupation and benefits of implanting a permanent pacemaker. Ultimately, it was decided he would not be a candidate for a permanent pacemaker, and a watch-and-wait strategy was adopted.
Discussion: The autonomic nervous system is the key regulator of our rhythm during sleep. The sleep stages are divided into rapid eye movement (REM) and non-REM stages, each stage with a unique autonomic profile. REM sleep is characterised by increases in sympathetic activity, whereas in non-REM sleep there is an increase in parasympathetic tone and decrease in sympathetic influx. Commonly, bradyarrhythmia during sleep may also be a manifestation of sleep apnoea syndromes (SAS) including OSA. Cannabis use has been linked to symptomatic bradycardia; a case series of symptomatic bradycardia in young cannabis users showed that cessation of cannabis use helped resolve bradycardia. Cannabinoids are thought to act centrally by enhancing parasympathetic response and damping the sympathetic drive. The syndrome of REM sleep-related bradycardia is another explanation for nocturnal bradycardia. First described in 1984, this rare clinical entity was characterised by transient sinus arrest or complete heart block with ventricular arrest during the REM phase, independently of apnoea. A case report by Duba et al. described a patient with symptomatic REM sleep bradycardia who underwent electrophysiology study (EPS), which showed sinus nodal disease, and subsequently received a dual chamber pacemaker. European guidelines suggest that EPS may be considered in patients with syncope in whom bradycardia is suspected, but it is not routinely recommended for bradyarrhythmias.
Conclusion: The case highlights the difficulty in deciding optimum management for nocturnal bradyarrhythmias, particularly those that are symptomatic and do not fit what is considered as ‘benign’ physiological response to sleep and not related to sleep apnoea. The role of invasive EPS and permanent pacemaker implantation is uncertain and not included in guidelines. Hence, nocturnal bradyarrhythmias with negative sleep study for SAS is an area of electrophysiology that still warrants more research. ❑