Introduction: There are few data on strategies to improve access to complex cardiac implantable electronic device (CIED) implantation and other treatments for heart failure (HF) or left ventricular systolic dysfunction (LVERSUSD). We hypothesized that a cohort of patients in primary care would already qualify for a CIED but had not received an implant. We set out to identify patients within primary care with HF/LVERSUSD requiring optimisation of their current HF treatment through optimisation of medical therapy or complex CIED therapy.
Methods: All relevant medical records were audited in GP practices to assess current management, compliance with NICE guidelines and suitability for a complex CIED implant. Audit review focused on symptoms that may be compatible with HF, those with a history of myocardial infarction, percutaneous or surgical coronary intervention or atrial fibrillation, the last echocardiogram/ECG, ejection fraction ≤35% or moderate severe LVSD and use of prognostic HF drugs. The case finder element of GRASP-HF was used to identify patients with HF / LVERSUSD that had not been correctly coded in GP records. Patients identified as suitable for clinical review were invited to attend face to face consultations at their local GP practice for an up to date ECG and where consultant cardiologists with a specialist interest in CIED formed a management plan. If needed, patients were referred by the cardiologist into secondary/tertiary care for CIED management and/or treatment optimisation. Outcomes after each patient clinical review were assessed up to 12-months.
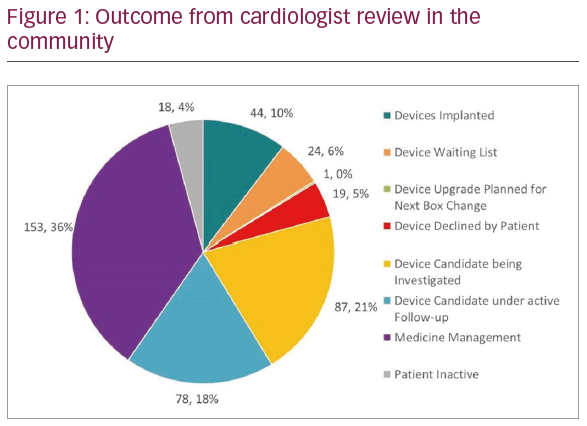
Results: HF audits were carried out between January 2018–April 2019 across 42 GP practices covering the north west, midlands, and south west of England (population: 520,854 patients). Total combined HF register was 5,842. A further 5,740 patients were identified by the GRASP-HF element. From 11,582 patients audited, 424 patients invited for a cardiologist review in the community. Following first clinical review, 26 (6.1%) patients were deemed suitable for urgent CIED implantation, 208 (49.1%) were classified as potential CIED candidates requiring both medical optimisation and repeat assessment of LV function, 91 (21.5%) required medicines optimisation only, and 99 (23.3%) did not have any change in their management. After follow-up, 88 patients were deemed appropriate for CIED implantation (with 44 Implanted, 24 on waiting list, one scheduled for upgrade at next box change, and 19 who declined intervention). All outcomes for the 424 patients are shown in Figure 1. 16.2% of all patients reviewed either received a CIED implant or are on a waiting list for this (27 CRT-P, 20 ICD, and 22 CRT-D).
Conclusion: Focused audits within primary care are an effective and efficient way of identifying patients who qualify for a complex CIED and/or optimisation of medical therapy.