Introduction: Patients with congenital heart disease (CHD) are predisposed to a range of cardiac arrhythmias. This may be a result of the primary congenital lesion or a result of ensuing surgical or percutaneous interventions. Implantation of cardiac rhythm management (CRM) devices can be of huge benefit both symptomatically and prognostically, however the implant procedure and subsequent follow-up often provides additional challenges not seen in the non-CHD population.
Methods: This specialist CHD CRM clinic was established in April 2018 with the intention of improving patient outcomes and continuity of care, whilst also streamlining clinical efficiency. One-hundred-and-fifty-four CHD patients were identified as having active appointments booked with the CRM clinic. At 14 months 65% (100) of the total number of CHD-CRM patients had been seen at least once in the specialist clinic. The average number of patients seen in April–December (inclusive) 2018 per month was 12.6, increasing to 22.4 between January and May 2019. Outcomes were evaluated by four criteria: no changes/medical review needed, major programming changes, minor programming changes and need for medical review. A need for medical review was recorded whenever a conversation with the consultant was documented, or where a change in medication was initiated. Major programming changes were classified as ≥ three changes or a change of pacing mode or tachycardia therapy. Minor programming changes were recorded where ≤ three changes were made. The results were broken into three separate time periods; April-December 2018, January–March 2019, and April–May 2019, and calculated as a percentage of the total number of outcomes. For comparison, outcomes were also recorded for 50 patients seen outside of the CHD-CRM clinic.
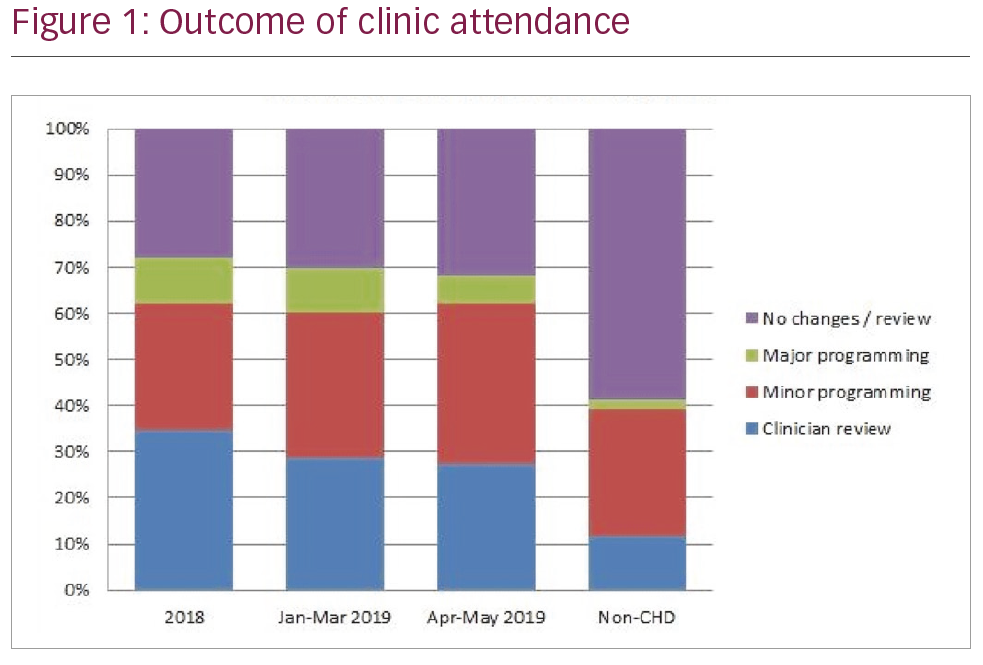
Results: Patients with CHD were observed to have different outcomes at CRM follow-up appointments than seen in the standard clinic. In the most recent data set, 21 patients (31%) in the CHD CRM clinic required no further clinical input or programming changes compared to 30 (59%) in the standard clinic. The number of major programming changes was four (6%) compared with one (2%), and minor programming changes 23 (35%) compared to 14 (27%). A clinician review was required for 18 (27%) patients with CHD, but only six (12%) from the non-CHD population. These findings illustrate the complex and clinically challenging nature of follow-up for these patients and the increased burden on services that results. By seeing these patients in a dedicated clinic, we were able to streamline access to medical input and reduce the total number of reviews and programming changes that were necessary with a reduction in episodes of clinician input from 35% in 2018 to 27% in recent months. Activity in the clinic continues to increase. Of note, an increased tariff is applicable to the clinic as a specialist service.
Implications: Further work is needed to quantitatively and qualitatively assess clinical outcomes at a patient level, along with service efficiency and patient satisfaction. It is anticipated that this service will deliver more efficient and clinically superior care for this sub-group of patients, as well as a platform for further research and review of CRM best-practice in the CHD population. It may also provide unique training opportunities for physiologists with an interest in CHD.