Background: Subcutaneous implantable cardioverter defibrillators (S-ICD) have been implanted at the Nottingham University Hospitals NHS Trust since December 2014, by a single operator with no prior experience of implant technique, with a proctor for two cases.
Objective: To report on demographics of patients selected for the S-ICD; procedural data and success, and complication rates.
Methods: A database of all new S-ICD implants over the past 4.5 years was analysed and compared to the post-market approval study (PAS), which was performed in a contemporary real-world setting.1 Procedural success was measured as conversion rate of induced ventricular fibrillation. System- and procedure-related complications were defined as complications that were caused by, or would not have occurred in the absence of the S-ICD. Complications were considered according to procedure volume (Quartile (Q) 1: implants 1–4; Q2: implants 5–12; Q3: implants 13–28; Q4: implants >28), recognising the learning curve associated with a new procedure,2 and were reported at 30 and 180 days.
Results: From December 2014 to May 2019, 57 patients were implanted. The aetiology was Brugada syndrome (14%); idiopathic ventricular arrhythmia (22.8%); long QT syndrome (7%); ischaemic cardiomyopathy (10.5%); dilated cardiomyopathy (15.8%); hypertrophic cardiomyopathy (10.5%); other (12.3%).
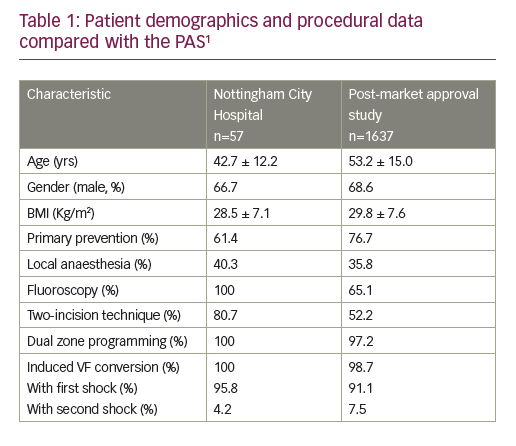
Table 1 shows patient demographics and procedural data compared to the PAS. Our patients were younger and had less comorbidities (chronic kidney disease [7%versus 25.6 %]; diabetes [3.5% versus 33.6%]; ischaemic heart disease [17.5% versus 33.2%]; atrial fibrillation [15.8% versus 16.2%]; LVEF ≤35% [30.2% versus 75.4%]).
We used fluoroscopy for all procedures, and performed 40% of our procedures under local anaesthesia. After the initial learning curve, the two-incision technique was used preferentially. Ventricular fibrillation was induced in 48 cases, and all were successfully converted. All patients had dual zone programming.
Compared to the study by Knops et al. which involved 889 patients,2 our 180-day complication rates for Q 1 were 50 % versus 9.8% (two patients had sub-optimal electrode position, one of whom underwent the procedure with a proctor); Q 2: 0% versus 6.4%; Q 3: 18.8% versus 5.1%; and Q 4: 4.5% versus 5.4%. Compared to the PAS,1 our 30-day complication rates for Q 4 were lower at 3.4% versus 3.7%.
Conclusion: We appeared to have more stringent selection criteria for the S-ICD compared to the PAS1, implanting in a younger population with lesser comorbidities and a high incidence of inherited cardiac conditions. Our procedural success was higher, with lower complication rates after the initial learning curve compared to published data.1,2
References
1. Gold M, Aasbo JD, El-Chami MF, et al. Subcutaneous implantable cardioverter-defibrillator Post-Approval Study: Clinical characteristics and perioperative results. Heart Rhythm. 2017;14(10):1456–63.
2. Knops RE, Brouwer TF, Barr CS, et al. The learning curve associated with the introduction of the subcutaneous implantable defibrillator. Europace. 2016;18(7):1010–5.







