Nearly half of patients who present with angina have non-obstructed coronary arteries (ANOCA).1 A significant proportion of these patients have a coronary vasomotor dysfunction, such as coronary microvascular disease and/or coronary artery spasm.2,3 Vasospastic angina (VSA) refers to a dysfunctional state where there is sudden coronary flow attenuation as a result of either epicardial or microvascular spasm, leading to downstream myocardial ischaemia and angina. This phenomenon was first reported by Prinzmetal et al., who described it as a distinct pathology that leads to resting chest tightness associated with marked ST-segment elevation and a much greater prevalence of ventricular arrhythmias than classical effort angina.4 The authors termed this entity ‘variant angina’ and hypothesized that it occurs due to a sudden and transient increase in vessel tone. Coronary artery spasm is now widely recognized as a distinct pathophysiological entity that can lead to myocardial ischaemia. In 2015, the Coronary Vasomotion Disorders International Study (COVADIS) group proposed the international standardization of diagnostic criteria for VSA to harmonize clinical and research work on VSA.5 This allows ‘definitive VSA’ to be diagnosed either by coronary angiography with pharmacological stimulation and/or by ambulatory electrocardiogram (ECG) recording. Both of these approaches are recommended by the European Society of Cardiology and the Japanese Circulation Society when investigating patients with suspected VSA.6,7
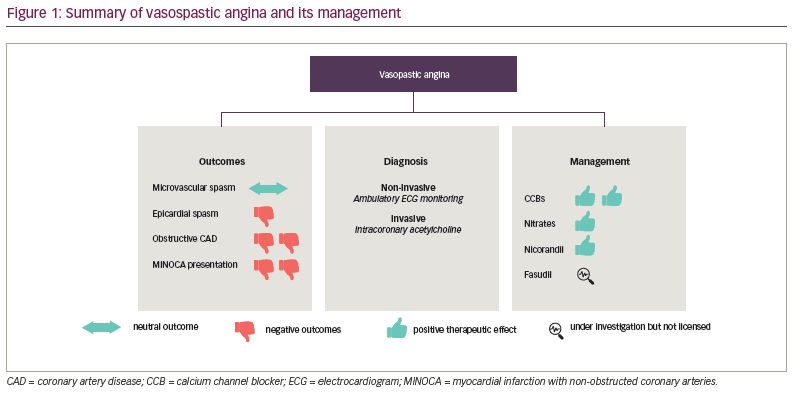
VSA has been associated with poor quality of life and increased risk of adverse cardiovascular outcomes.8 However, its underlying pathophysiology remains incompletely defined, and the factors contributing to the development of VSA are poorly understood. The current management of VSA focuses on mitigating cardiovascular risk factors and anti-anginal therapy, specifically calcium channel blockers (CCBs) and long-acting nitrates. The focus of this review article will be on the pathophysiology, diagnosis and contemporary management of patients with VSA (Figure 1).
Flow augmentation in the healthy vasculature
The coronary vasculature comprises epicardial arteries (>400 μm), pre-arterioles (100–400 μm), arterioles (<100 μm) and capillaries (<10 μm).9 The epicardial arteries function as conduit vessels. The arterioles regulate coronary vascular resistance and, therefore, coronary blood flow (CBF) in response to changes in myocardial oxygen demand.9 The capillary bed delivers oxygen and substrates to the myocytes.9 The endothelium plays an important role in the modulation of vascular tone by synthesizing and releasing several vasodilator substances, such as nitric oxide (NO).9 Increased endothelial wall shear stress and acetylcholine (ACh) are determinants of CBF in health.9
The pathophysiology of vasospastic angina
Coronary endothelial dysfunction
In the presence of functional endothelium, the balance of shear stress-induced vasodilation and vasoconstriction tips towards the former; however, in the presence of endothelial dysfunction, the balance tips towards the latter.10,11 Under normal physiological circumstances, shear stress, by activating mechanoreceptors on endothelial cells, triggers the endothelial NO synthase (eNOS), in the presence of its cofactor tetrahydrobiopterin, to convert L-arginine into NO.11 However, certain conditions, such as a systemic inflammatory state, impair the ability of eNOS to produce NO; this is known as ‘eNOS uncoupling’.11 Coronary endothelial dysfunction is thought to be a precursor of obstructive coronary artery disease (CAD), and it is a marker of adverse cardiovascular outcomes.12 In the setting of coronary artery spasm, several clinical studies have demonstrated reduced NO activity.13 Furthermore, the observation that animal models with mutations of the eNOS gene are predisposed to developing coronary artery spasm further supports the contribution of coronary endothelial dysfunction in the pathogenesis of coronary artery spasm.14 ACh is used as the preferred agent to test coronary endothelial integrity in the catheter laboratory; this is because of its dual action on muscarinic receptors on the endothelium and vascular smooth muscle (VSM).
Dysfunctional endothelial cells release endothelin-1 (ET-1), which is a potent vasoconstrictor. Several clinical studies have demonstrated higher coronary sinus plasma ET-1 levels in patients with demonstrable coronary artery spasm during provocation assessment.15 Ford et al. reported an attenuated vasorelaxation in response to ACh and augmented vasoconstrictive response to ET-1 in gluteal biopsy samples of patients with VSA compared with control subjects, indicating a state of systemic endothelial dysfunction in these patients.16 Finally, in a cohort of patients with ANOCA, Reriani et al. reported an improvement in coronary endothelial function after treatment with an endothelin A receptor antagonist.17
However, whilst coronary endothelial dysfunction has been implicated in the development of coronary artery spasm, it has been demonstrated that not all vessels that are predisposed to spasm have underlying coronary endothelial dysfunction.18 This suggests that there may be an additional mechanism that, in the presence (or sometimes even in the absence) of coronary endothelial dysfunction, leads to coronary artery spasm. VSM hyperreactivity may represent this additional mechanism and is discussed below in detail.
Vascular smooth muscle hyperreactivity
Although the mechanisms leading to VSM hyperreactivity are not fully understood, it is thought to be a manifestation of an alteration of the signal transduction pathway somewhere between, but not including, the cellular receptors and the contractile proteins in the VSM cell. Porcine models of coronary spasm have demonstrated that the calcium handling mechanism of contractile proteins remains unaltered, as does the expression of cellular receptors involved in promoting vasoconstriction.19,20 Animal studies have also implicated the protein kinase C-mediated pathway in the pathogenesis of coronary artery spasm.21 These results suggest that calcium (Ca2+) entry through L-type Ca2+ channels into VSM cells is the initial trigger for coronary artery spasm and that Ca2+ entry might be augmented via protein kinase C-dependent mechanisms. Indeed, it has been demonstrated that L-type Ca2+ channels are functionally upregulated at the spastic site in a porcine model of coronary artery spasm.22 Animal studies have also reported that rho kinase is upregulated at the spastic site and plays a key role in inducing VSM hypercontraction by inhibiting myosin light chain phosphatase.23 Fasudil, a rho kinase inhibitor, has been shown to markedly attenuate ACh-induced coronary vasoconstriction in patients with coronary artery spasm.24 It has been hypothesized that coronary endothelial dysfunction plays a greater role in diffuse multi-vessel spasm, whereas VSM hyperreactivity plays a greater role in focal spasm.
Clinical presentation and outcomes
VSA should be suspected in patients with anginal symptoms occurring predominantly at rest, especially if the resting symptoms follow a diurnal pattern (being worse at night and in the early morning). Although Prinzmetal’s reports had linked episodes of coronary vasospasm predominantly with ST-segment elevation, there is now a greater appreciation that an episode of coronary artery spasm can present with disparate ischaemic ECG changes commensurate with the degree of coronary flow attenuation.4 Prolonged and more occlusive episodes of coronary artery spasm have a greater propensity to lead to ventricular arrhythmias; this is thought to be due to an increased inhomogeneity of ventricular depolarization and repolarization due to acute, severe and transient myocardial ischaemia.25 These factors increase ventricular vulnerability and heighten the risk of sudden cardiac death.26
The major adverse cardiac events rate, a composite of death, non-fatal myocardial infarction (MI), unstable angina and heart failure, has been reported to be around 5–6% over a median follow-up period of 3–4 years in patients with VSA.27,28 Two endotypes of coronary artery spasm are recognized: epicardial spasm (arbitrarily defined as ≥90% epicardial artery vasoconstriction in response to ACh stimulation, ischaemic ECG changes and characteristic chest pain) and microvascular spasm (a diagnosis of exclusion, namely <90% epicardial artery vasoconstriction in response to ACh stimulation, ischaemic ECG changes and characteristic chest pain).5 A recent study reported a 7.5% incidence of all-cause mortality, 1.4% MI and 2.2% stroke over a median 7-year follow-up in patients with invasively characterized coronary artery spasm.8 Recurrent symptoms were reported in 64% of patients, and 12% of patients underwent a repeat coronary angiography. Multivariate analysis revealed epicardial spasm as a predictor of non-fatal MI and repeat angiography, whereas patients with microvascular spasm more often had recurrent angina at follow-up.8 Whilst the overall prognosis of patients with ANOCA and coronary artery spasm is generally favourable, patients with obstructive CAD who are predisposed to spasm have a worse outlook.29 Furthermore, patients with obstructive CAD who develop spasm within the stenotic segment are more likely to suffer from adverse cardiovascular outcomes compared with those who develop spasm in the non-stenotic coronary segment or those who do not develop spasm at all.30 The mechanisms underlying this are unclear; however, animal studies have demonstrated that intimal injury is prevalent in stenotic segments that develop spasm with pharmacological stimulation.31 Therefore, it is conceivable that spasm within a stenotic segment can cause plaque disruption and, therefore, predispose to acute coronary syndrome. Finally, patients with MI with non-obstructed coronary arteries secondary to coronary vasospasm have a heightened risk of all-cause mortality, cardiac death and readmission with acute coronary syndrome.32,33 Coronary artery spasm provocation assessment was shown to be safe in patients presenting with MI with non-obstructed coronary arteries, and it helps identify a high-risk patient cohort who may benefit from close follow-up and aggressive pleiotropic and anti-ischaemic therapies.32,33
Diagnosis
Non-invasive assessment of coronary vasospasm
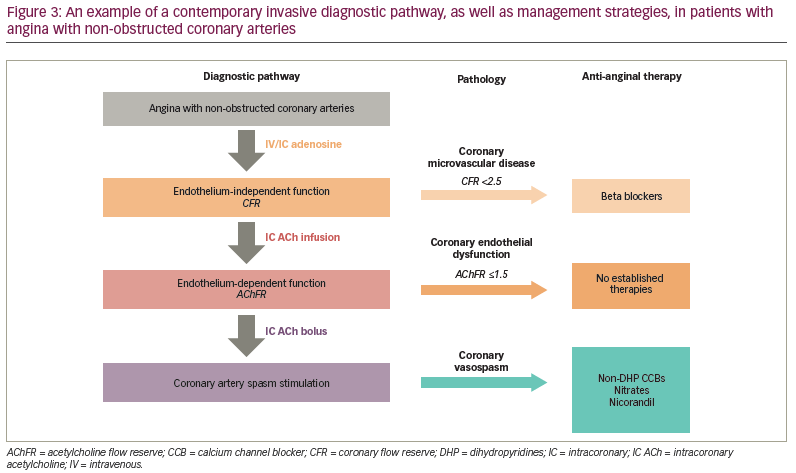
According to the COVADIS criteria, if a spontaneous episode of rest angina is associated with transient ischaemic ECG changes, and if there is no other cause identified for the ECG changes, then coronary artery spasm is presumed to be responsible, and a definitive diagnosis of VSA may be made without formal documentation of coronary artery spasm.5 However, it is generally not feasible to document ischaemic ECG changes during spontaneous episodes of rest angina. Furthermore, coronary artery spasm frequently co-exists with epicardial CAD and/or coronary endothelium-independent dysfunction. Therefore, performing one procedure (i.e. coronary angiography with invasive coronary physiology assessment) that allows for a comprehensive assessment of the entire coronary vascular function (fractional flow reserve, coronary flow reserve [CFR], ACh flow reserve [AChFR] and spasm assessment) and provides insights into the spasm endotype (epicardial versus microvascular spasm) is now the investigation of choice in this patient cohort.34
Invasive coronary physiology assessment in the catheter laboratory
Coronary vascular assessment with ACh stimulation can be performed readily and safely in patients with suspected VSA.35 It is recommended that patients with ANOCA should undergo coronary physiology assessment to detect coronary vascular dysfunction that can act as a substrate for myocardial ischaemia. The main parameter used to distinguish normal from abnormal coronary vascular function is the CFR. CFR is the ratio of hyperaemic to baseline CBF in response to adenosine and reflects the ability of the coronary vasculature to augment blood flow in response to increased demand. An impaired CFR, defined as CFR <2.5, in the presence of a normal fractional flow reserve is suggestive of endothelium-independent coronary microvascular disease; this is associated with an increased likelihood of myocardial ischaemia on non-invasive assessment and adverse cardiovascular outcomes.34,36,37
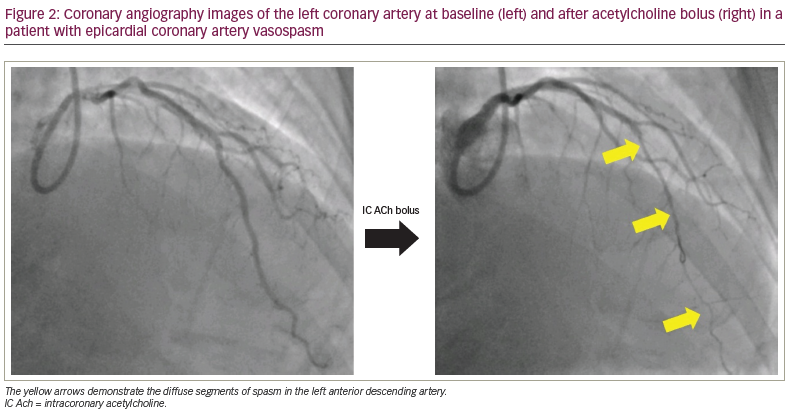
Intracoronary ACh infusion, at concentrations of up to 10-4 mol/L, can be used to assess coronary endothelial function, with a normal response being an increase in CBF by 50% or more compared with the basal flow (i.e. an AChFR of >1.5).38 An AChFR of ≤1.5 has been associated with myocardial ischaemia on non-invasive assessment and with an increased risk of adverse outcomes.12,38,39 In cases where the pre-test probability of coronary vasospasm is high, operators should carry out coronary spasm assessment using ACh bolus.34 There is variation in the doses and delivery rates of ACh used during spasm assessment, although the underlying scientific rationale remains the same. The general consensus is to deliver a 100 µg bolus of ACh down the left anterior descending artery over 20 seconds; this dose needs to be halved (i.e. 50 µg over 20 seconds) if being delivered into the right coronary artery due to the higher risk of bradyarrhythmias.34 Clinicians should remain vigilant and promptly manage ACh-induced tachyarrhythmias with pharmacological/electrical cardioversion and spasms with intracoronary nitroglycerin. A diagnosis of epicardial artery spasm is made when ACh bolus leads to ≥90% coronary vasoconstriction, ischaemic ECG changes and chest pain; this protocol and diagnostic threshold is associated with a high degree of sensitivity and specificity for the detection of coronary spasm in patients with VSA symptoms.5,40 The diagnosis of microvascular spasm is made when ACh bolus leads to ischaemic ECG changes and chest pain in the absence of ≥90% coronary vasoconstriction; in the absence of significant epicardial spasm, an AChFR <1.0 with ACh bolus is also suggestive of microvascular spasm as it demonstrates flow attenuation.5,34 An example of a vessel with significant epicardial spasm in response to ACh bolus is shown in Figure 2.
Diagnostic challenges
Whilst our suggested ACh protocol is the most commonly used one, other derivations exist and are associated with varying degrees of sensitivity and specificity.34 These include the incremental infusion of ACh at 0.86, 8.63, 86.3, 863 µg/mL over 3 minutes or incremental boluses of ACh at100–200 µg over 20 seconds.7,41 These derivations have important clinical implications; for example, a 200 µg bolus is more likely to lead to multivessel spasm than a 100 µg bolus, and a 20-second bolus is more likely to lead to vasospasm than a 3-minute infusion of the same dose.42,43 Furthermore, it is not known what dose and infusion rate of ACh correlates with physiological degrees of spasm, the caveat being that, beyond a certain threshold of dose and infusion rate, ACh may provoke spasm in any individual.44 This phenomenon was observed in a study investigating the effects of varying concentrations of ACh in patients with normal epicardial arteries.45 The authors reported an increase in epicardial vessel diameter and CBF with ACh concentrations of up to 10-4 mol/L; however, significant vasoconstriction, accompanied by chest pain, was observed with concentrations of 10-3 mol/L.45 This led the authors to conclude that the local ACh concentration and the coronary vascular segment under question may play a significant role in the observed response to ACh.45 Finally, the diagnostic threshold for the degree of epicardial vasoconstriction in response to ACh can also vary between centres. The majority of centres use the 90% threshold; however, some centres use different arbitrarily chosen thresholds, such as 75% vasoconstriction.34,35 Using different diagnostic thresholds will, of course, alter the diagnostic sensitivity and specificity.
Therefore, although invasive coronary physiology assessment with ACh stimulation remains the investigation of choice in patients with suspected VSA, it has certain caveats that clinicians should bear in mind and assert their discretion when necessary. Furthermore, invasive spasm assessment should be reserved only for patients with clinical symptoms suggestive of VSA.
Management
The mainstay of management of VSA is pharmacological, recommendation of lifestyle changes (such as smoking cessation) and avoidance of agents that can provoke coronary spasm (such as beta blockers and triptans). We discuss some of the commonly used anti-ischaemic agents used in these patients.
Calcium channel blockers
CCBs inhibit the voltage-dependent L-type Ca2+ channels and, therefore, reduce calcium–calmodulin-induced myosin light chain kinase activation. CCBs suppress inducible coronary spasm and, therefore, lead to reduced angina frequency and improved prognosis.46–50
Long-acting nitrates
Nitrates act as NO donors, resulting in vasodilation. They have a disparate mechanism of action compared with CCBs; therefore, patients can be treated with a combination of a CCB and nitrate to target separate pathways of coronary spasm. Nitrates are also effective in reducing anginal episodes.51
Nicorandil
Nicorandil leads to vasodilation via two mechanisms. Firstly, it stimulates soluble guanylate cyclase and leads to increased cyclic guanosine monophosphate concentrations. Secondly, it leads to hyperpolarization by opening the adenosine triphosphate-sensitive potassium channels; this subsequently leads to a closure of the calcium channels. Nicorandil reduces anginal burden in patients with VSA.52 The Japanese Cardiology Society give a IIa recommendation for the use of nicorandil in patients with VSA, although it remains the second line in European guidelines.6,7
Rho kinase inhibitors
Clinical studies have demonstrated the efficacy of fasudil, a rho kinase inhibitor, in ameliorating coronary artery spasm induced by ACh.24,53 The myosin-binding substrate promotes vasodilation by dephosphorylating the myosin head and causing the detachment of the myosin–actin crosslink. However, rho kinase inhibits myosin-binding substrate and, therefore, promotes a vasoconstrictive state. By inhibiting this action, rho kinase inhibitors promote a vasodilatory state. However, this agent is not available outside of Japan for clinical use at present.
All of these agents target key cellular pathways in the coronary vasomotor regulation, with CCBs targeting the L-type calcium channels, long-acting nitrates acting as NO donors, nicorandil promoting cyclic guanosine monophosphate production, and endothelin receptor antagonists and rho kinase inhibitors dampening endothelin and rho kinase-dependent vasoconstrictive pathways.
Pleiotropic agents
In patients with demonstrable coronary endothelial dysfunction, angiotensin-converting enzyme inhibitors and statins may be used, as there is evidence that these agents enhance coronary endothelial function through a reduction in oxidative stress. A randomized open-label study comparing 6 months of fluvastatin and CCB (combination therapy) versus CCB alone demonstrated that the combination therapy led to a greater amelioration in the development of ACh-induced spasm compared with CCB therapy.54 Furthermore, whilst the inflammatory biomarkers were matched between the two groups at baseline, patients in the combination therapy arm had significantly lower C-reactive protein levels at the end of the study, whereas there was no change in patients in the CCB arm.54 Finally, in a large registry-based propensity-matched comparison study, the prevalence of recurrent angina, major adverse cardiac events and death at 5 years was lower in patients with VSA who were taking angiotensin-converting enzyme inhibitors versus those who were not.55
The Coronary microvascular angina (CorMicA ; ClinicalTrials.gov identifier: NCT03193294) study has demonstrated that stratifying treatment in patients with ANOCA based upon coronary vascular physiology assessment yields superior outcomes to empirical therapy, supporting the role of comprehensive coronary physiology testing in this patient cohort.3,56 Furthermore, a recent study has demonstrated the ability to predict patients’ responses to nitrates by giving intracoronary nitrates to those with demonstrable spasm on invasive assessment, followed by a rechallenge with a second dose of ACh.57 The authors reported that nitrates attenuated epicardial vasospasm in most patients, whereas this desired response was less frequently observed in patients with microvascular spasm. This is an example of nuanced personalized therapy that may lead to better patient outcomes, and this protocol can be used as a template to assess individual responses to anti-ischaemic agents in the catheter laboratory in order to select the most efficacious medication for a given individual.57 Finally, following on from the findings that the endothelin pathway is implicated in patients with VSA,16 the Precision medicine with zibotentan in microvascular angina (PRIZE; ClinicalTrials.gov identifier: NCT04097314) study is investigating whether an endothelin receptor antagonist (zibotentan) can improve patient-centric outcomes in patients with coronary microvascular disease and VSA.58
An example of a contemporary diagnostic pathway and management options is shown in Figure 3.
Conclusion
Coronary artery spasm leading to VSA is common in patients presenting with ANOCA and is associated with poor quality of life and adverse cardiovascular outcomes.
VSA can be diagnosed accurately and safely in the catheter laboratory. The mainstay of management is pharmacological, with CCBs and long-acting nitrates being the first-line therapies, and nicorandil being second line. Other therapies targeting the pertinent mechanistic pathways have shown promise in clinical trials. There is now growing evidence that nuanced personalized therapy may be associated with better patient-centric outcomes than empirical therapy in these patients.
Future directions
The main challenge for the field during the next few years will be to ensure appropriate uptake of invasive intracoronary assessment to fully evaluate the coronary vasculature in patients with ANOCA and high pre-test probability of VSA. Further mechanistic work needs to be carried out to develop novel therapeutic options that target specific pathways within the endothelial and VSM cells to personalize patient care.